Abstract
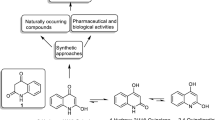
We consider different variants for synthesis of 2-R-oxazolo[4,5-c]quinolin-4(5H)-ones based on 3-amino-1H-2-oxo-4-hydroxyquinolines and their 3-N-acyl derivatives. We show, that in the latter case, formation of the oxazole ring is possible via two routes, depending on the nature of the substituent on the acyl residue.
Similar content being viewed by others
References
I. V. Ukrainets, S. G. Taran, O. L. Kodolova, O. V. Gorokhova, and V. N. Kravchenko, Khim. Geterotsikl. Soedin., No. 8, 1100 (1997).
P. Albaugh, US Pat. 5, 182,290; Ref. Zh. Khim., 3O66P (1996).
R. C. Elderfield (ed.), Heterocyclic Compounds, Vol. 5 [Russian translation], Inostr. Lit., Moscow (1961).
I. V. Ukrainets, S. G. Taran, L. V. Sidorenko, O. V. Gorokhova, A. A. Ogirenko, A. V. Turov, and N. I. Filimonova, Khim. Geterotsikl. Soedin., No. 8, 1113 (1996).
V. V. Mezheritskii, E. P. Olekhnovich, S. M. Luk'yanov, and G. N. Dorofeenko, Ortho Esters in Organic Synthesis [in Russian], Izdat. Rost. Univ., Rostov-na-Donu (1976).
I. V. Ukrainets, S. G. Taran, L. V. Sidorenko, O. V. Gorokhova, A. V. Turov, and A. A. Ogirenko, Khim. Geterotsikl. Soedin., No. 7, 933 (1997).
Additional information
For Communication 35, see [1].
Ukrainian Pharmaceutical Academy, Kharkov 310002. Translated from Khimiya Geterotsiklicheskikh Soedinenii. No. 11. pp. 1536–1541, November, 1997.
Rights and permissions
About this article
Cite this article
Ukrainets, I.V., Taran, S.G., Sidorenko, L.V. et al. 4-Hydroxy-2-quinolones. 36. Synthesis of 2-R-oxazolo[4,5-c]quinolin-4(5H)-ones. Chem Heterocycl Compd 33, 1328–1333 (1997). https://doi.org/10.1007/BF02320336
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02320336