Abstract
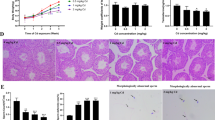
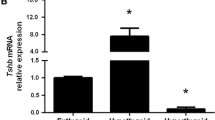
The present study aimed at investigating whether previously reported interstrain differences of mice in testicular susceptibility to cadmium toxicity can be attributed to different metallothionein expression in the testes and other organs. Furthermore, purified Leydig cell systems were employed to identify a testicular compartment capable to respond specifically to exogenous cadmium by increased metallothionein expression. It was shown that species differences in testicular damage after cadmium administration can not be explained by differences in cadmium-induced stimulation of hepatic and renal metallothionein. In contrast to these organs, no metallothionein response to cadmium was observed in testicular homogenates. However, both freshly isolated and purified rat Leydig cells and a murine tumor Leydig cell line are not only able to synthesize metallothionein under basal conditions, but also respond to exogenous cadmium by increased synthesis of metallothionein (35S-cysteine incorporation) and an increased Mt-mRNA content. Extracts from freshly isolated and purified rat Leydig cells contained a cadmiumbinding component which eluted in gel chromatography with the same properties as metallothionein. It was concluded that the use of purified cell systems is a valuable tool to investigate possible targets of cadmium toxicity.
Similar content being viewed by others
References
Abel J, Hoehr D, Schurek HJ (1987) Renal handling of cadmium and cadmium-metallothionein: studies on the isolated perfused rat kidney. Arch Toxicol 60: 370–375
Andersen RD, Piletz JE, Birren BW, Herschman HR (1983) Levels of metallothionein messenger RNA in foetal, neonatal and maternal rat liver. Eur J Biochem 131: 497–500
Banerjee D, Onosaka S, Cherian MG (1982) Immunohistochemical localization of metallothionein in cell nucleus and cytoplasm of rat liver and kidney. Toxicology 24: 95–105
Bhave RM, Wilson MJ, Waalkes MP (1988) Methylation status and organization of the metallothionein-I gene in livers and testes of strains of mice resistant and susceptible to cadmium. Toxicology 50: 231–245
Chellman GJ, Shaikh ZA, Baggs RR (1984) Decreased uptake and altered subcellular disposition of testicular cadmium as possible mechanism of cadmium resistance to cadmium-induced testicular necrosis in inbred mice. Toxicology 30: 157–169
Chellman GJ, Shaikh ZA, Baggs RR, Diamond GL (1985) Resistance to Cadmium-induced necrosis in testes of inbred mice: possible role of metallothionein-like cadmium-binding protein. Toxicol Appl Pharmacol 79: 511–523
Chen RW, Ganther HE (1975) Some properties of a unique cadmiumbinding moiety in the soluble fraction of rat testes. Environ Physiol Biochem 5: 235–243
Chiquoine AD, Suntzeff V (1965) Sensitivity of mammals to cadmium necrosis of the testis. J Reprod Fertil 10: 455–457
Chirgwin JM, Przybyla AE, MacDonald RJ, Rutter WJ (1979) Isolation of biologically active ribonucleic acid from source enriched in ribonuclease. Biochemistry 18: 5294–5299
Cox DR, Palmiter RD (1983) The metallothionein-I gene maps to mouse chromosome 8: Implication for human Menkes' disease. Hum Genet 64: 61–64
Danielson KG, Bencker L, Lindgren A (1982a) Immunochemical localization of metallothionein in rat liver and kidney. J Histochem Cytochem 30: 1033–1039
Danielson KG, Ohi S, Huang PC (1982b) Immunochemical detection of metallothionein in specific epithelial cells of rat organs. Proc Natl Acad Sci USA 79: 2301–2304
Dieter HH, Müller L, Abel J, Summer KH (1986) Determination of Cd-thionein in biological materials: comparative standard recovery by five current methods using protein nitrogen for standard calibration. Toxicol Appl Pharmacol 85: 380–388
Durnam DM, Palmiter RD (1981) Transcriptional regulation of the mouse metallothionein-I gene by heavy metals. J Biol Chem 256: 5712–5716
Eaton DL, Toal BF (1982) Evaluation of the Cd/hemoglobin affinity assay for the rapid determination of metallothionein in biological tissues. Toxicol Appl Pharmacol 66: 134–142
Glissin V, Crkvenjakow R, Byus C (1974) Ribonucleic acid isolated by cesium chloride centrifugation. Biochemistry 13: 2633–2637
Gunn SA, Gould TC (1970) Specificity of the vascular system of the male reproductive tract. J Reprod Fertil Suppl 10: 75–95
Gunn SA, Gould TC, Andersen WAD (1965) Strain differences in susceptibility of mice and rats to cadmium-induced testicular damage. J Reprod Fertil 10: 273–275
Gunn SA, Gould TC, Andersen WAD (1968) Selectivity of organ response to cadmium injury and various protective measures. J Pathol Bacteriol 96: 89–96
Hamer DH (1986) Metallothionein. Annu Rev Biochem 55: 913–951
Hata A, Tsumoo H, Nakajima H, Shintaku K, Kimura M (1980) Acute cadmium intoxication in inbred mice: a study on strain differences. Chem Biol Interact 32: 29–39
Hunt DM, Mhlanga T (1983) Genetic studies on metallothionein synthesis in the mouse: the induction of metallothionein by cadmium in inbred strains. Biochem Genet 21: 609–625
Kägi JHR, Nordberg M (1979) Metallothionein. Birkhäuser, Basel
Kühn-Veiten N, Wolff J, Passia D, Staib W (1982) Determination of non-specific esterase activity in rat testis interstitial cells: a marker for evaluation of a Leydig cell purification procedure and of cell viability during incubation and continuous superfusion. Cell Mol Biol 28: 473–481
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of the bacteriophage T4. Nature 227: 680–685
Lowry OM, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193: 265–275
Lucis OJ, Lucis R (1969) Distribution of cadmium-109 and zinc-65 in mice of inbred strains. Arch Environ Health 19: 334–336
Mori H, Christensen AK (1980) Morphometric analysis of Leydig cells in the normal rat testis. J Cell Biol 84: 340–354
Mullin CH, Abel J, Klein D, Summer KH (1988) Metallothionein gene expression in mouse tissue by d-penicillamine. J Trace Elem Electrolytes Health Dis 2: 43–47
Nolan CV, Shaikh ZA (1986) An evaluation of tissue metallothionein and genetic resistance to cadmium toxicity in mice. Toxicol Appl Pharmacol 85: 135–144
Nordberg GF (1971) Effects of acute and chronic cadmium exposure on the testicles of mice. Environ Physiol 1: 171–187
Onosaka S, Cherian MG (1982) Comparison of metallothionein determination by polarographic and cadmium-saturation methods. Toxicol Appl Pharmacol 63: 270–274
Searle PF, Davison BL, Stuart GW, Wilkie TM, Norstedt G, Palmiter RD (1984) Regulation, linkage and sequence of mouse metallothionein I and II genes. Mol Cell Biol 4: 1221–1230
Taylor BA, Heiniger HJ, Meier H (1973) Genetic analysis of resistance to cadmium induced testicular damage in mice. Proc Soc Exp Biol Med 143: 629–633
Waalkes MP, Chernoff SB, Klaassen CD (1984a) Cadmium-binding proteins of rat testis: characteristics of a low-molecular mass protein that lacks identity with metallothionein. Biochem J 220: 811–818
Waalkes MP, Chernoff SB, Klaassen CD (1984b) Cadmium-binding proteins of rat testes: apperent source of the protein of low molecular mass. Biochem J 220: 819–824
Waalkes MP, Peratoni A, Palmer AE (1988) Isolation and partial characterization of the low-molecular-mass zinc/cadmium-binding protein from the testes of the patas monkey (Erythrocebus patas). Biochem J 256: 131–137
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Abel, J., de Ruiter, N. & Kühn-Veiten, W.N. Comparative study on metallothionein induction in whole testicular tissue and isolated Leydig cells. Arch Toxicol 65, 228–234 (1991). https://doi.org/10.1007/BF02307313
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02307313