Abstract
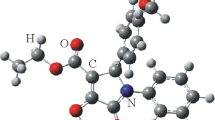
A simple procedure for synthesis of HIDA (2,6-dimethylphenylcarbamoylmethyl iminodiacetic acid) is described. It was obtained in better yield by reaction of 2,6-dimethylphenylcarbamoylmethyl chloride with iminodiacetic acid in carbon tetrachloride. The HIDA (A) was identified by IR and1H NMR spectroscopy. Two types of crystals were detected in the crystal final product. Since they could be separated, the crystals:A andB were investigated by X-ray analysis. Crystal and molecular structures were solved by direct methods and refined by full-matrix least-squares technique toR=0.047 andR=0.056, respectively. In the structure ofA, the N2 atom is protonated and the molecule exists as a zwitterion. The crystal structure of compoundA is stabilized by strong hydrogen bonds, which are reflected in higher close-packing coefficient, density, and melting point, compared to compoundB. CompoundB is a monomethyl ester of HIDA, which is present as an impurity in the final recrystallized product.
Similar content being viewed by others
References
Zmbova, B.; Konstatinovska-Djokic, D.; Tadzer, I.Int. J. Appl. Radiat. Isot. 1985,36, 389–394.
Zmbova, B.; Maksin, T.; Djokic, D.; Vladimirov, S.; Zivanov-Stakic, D.Isotopenpraxis 1990,26, 57–59.
Sheldrick, G. M.SHELX86. Program for Crystal Structure Determination; University of Göttingen, Federal Republic of Germany, 1986.
Sheldrick, G. M.SHELX76. Program for Crystal Structure Determination; University of Cambridge, England, 1976.
Jefrey, G. A. InProceedings of the Pre-Congress Symposium on Organic Crystal Chemistry; Poznan-Dymaczewo, 1978; p 102.
Kitaigorodskij, A. I.Molekulkristalle; Akademie-Verlag: Berlin, 1979; p 20.
Párkányi, L.; Kertesz, Cs.Computer Program for the Calculation of Molecular Volumes; Cent. Res. Inst. Chem.; Budapest, 1986.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ribár, B., Mészáros, C., Vladimirov, S. et al. Synthesis, crystal, and molecular structures of HIDA and its monomethyl ester. Struct Chem 6, 121–125 (1995). https://doi.org/10.1007/BF02281737
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02281737