Summary
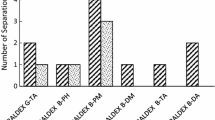
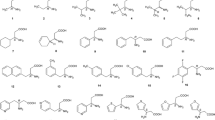
The resolution of nine recamic mixtures of tifluadom analogues has been evaluated using the chiral stationary phases Chiralcel OD and Chiralcel OJ. The separation was performed on an analytical scale to optimize the conditions for chiral resolution, approaching baseline separation, of the two enantiomers. Eight racemates were baseline separated on Chiralcel OJ using a mobile phase of hexane/2-propanol.
Similar content being viewed by others
References
R. L. Mueurisse, N. M. Blaton, O. M. Peeters, C. J. De Ranter, Acta Cryst. C48, 508 (1992); and references cited herein.
R. L. Meurisse, J.-Cl. Menier, C. J. De Ranter, submitted to Eur. J. Pharmacol.
D. Römer, H. H. Büscher, R. C. Hill, R. Maurer, P. I. Petcher, H. Zeugner, W. Benson, E. Finner, W. Milkowski, P. W. Thies, Life Sci.31, 1217 (1982).
R. S. L. Chang, V. J. Lotti, T. B. Chem, M. E. Keegan, Neurosci. Lett.72, 211 (1986).
M. G. Block, R. M. DiPardo, B. E. Evans, K. E. Rittle, W. L. Whitter, D. F. Veber, R. M. Freidinger, R. S. L. Chang, T. B. Chen, V. J. Lotti, J. Med. Chem.33, 450 (1990).
T. J. Petcher, A. Widmer, U. Maetzel, H. Zeugner, Acta Cryst.C41, 909 (1985).
E. J. Ariens, Eur. J. Clin. Pharmac.26, 663 (1984).
S. G. Allenmark, Chromatographic Enantioseparation: Methods and Applications, Ellis Horwood Ltd., Chichester (1988).
Y. Okamoto, R. Aburatani, Y. Kaida, K. Hatada Chem. Lett., 1125 (1988).
K.Ikeda, T. Hamasaki, H. Kohno, T. Ogawa, T. Matsumoto, J. Sakai, Chem. Lett., 1089 (1989).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Meurisse, R.L., De Ranter, C.J. Enantiomer separaton of tifluadom and analogues by liquid chromatography with cellulose-based chiral stationary phases. Chromatographia 38, 629–632 (1994). https://doi.org/10.1007/BF02277166
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02277166