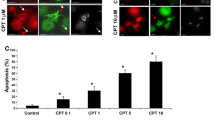
Summary
Pretreatment with cytochalasin B, which is known to disrupt microfilaments, significantly inhibits regulatory volume decrease (RVD) in Ehrlich ascites tumor cells, suggesting that an intact microfilament network is a prerequisite for a normal RVD response. Colchicine, which is known to disrupt microtubules, has no significant effect on RVD. Ehrlich cells have a cortical three-dimensional, orthogonal F-actin filament network which makes the cells look completely black in light microscopy following immunogold/silver staining using anti-actin antibodies. After addition of cytochalasin B, the stained cells get lighter with black dots localized to the plasma membrane and appearance of multiple knobby protrusions at cell periphery. Also, a significant decrease in the staining of the cells is seen after 15 min of RVD in hypotonic medium. This microfilament reorganization appears during RVD in the presence of external Ca2+ or Ca2+-ionophore A23187. It is, however, abolished in the absence of extracellular calcium, with or without prior depletion of intracellular Ca2+ stores. An effect of increased calcium influx might therefore be considered. The microfilament reorganization during RVD is abolished by the calmodulin antagonists pimozide and trifluoperazine, suggesting the involvement of calmodulin in the process. The microfilament reorganization is also prevented by addition of quinine. This quinine inhibition is overcome by addition of the K+ ionophore valinomycin.
Similar content being viewed by others
References
Albertini, D.F., Herman, B. 1984. Cell shape and membrane receptors dynamics modulation by the cytoskeleton.In: Cell and Muscle Motility: The Cytoskeleton. J.W. Shay, editor. Vol. 5, pp. 345–536. Plenum, New York, London
Appleman, M.M., Ariano, M.A., Takemoto, D.J., Whitson, R.H. 1982. Cyclic nucleotide phosphodiesterases.Hand. Exp. Pharmac. 58(1):261–300
Borisy, G.G., Taylor, E.W. 1967. The mechanism of action of colchicine.J. Cell Biol. 34:525–533
Chaldokov, G.N., Nabika, T., Nara, Y, Yamori, Y. 1989. Cyclic AMP- and cytochalasin B-induced arborization in cultured aortic smooth muscle cells: its cytopharmacological characterization.Cell Tissue Res. 255:435–442
Cheung, R.K., Grinstein, S., Dosch, H.M., Gelfand, E.W. 1982. Volume regulation by human lymphocytes: characterization of the ionic basis for regulatory volume decrease.J. Cell. Physiol. 112:189–196
Christensen, O. 1987. Mediation of cell volume regulation by Ca2+ influx through stretch-activated channels.Nature 330(5):66–68
Christensen, O., Hoffmann, E.K., Saermark, T., Sinonsen, L.O. 1988. Inositol triphosphate may be a second messenger in regulatory volume decrease in Ehrlich mouse ascites tumor cells.J. Physiol. 403 :109P
Christensen, O., Hoffmann, E.K. 1992. Cell swelling activates K and Cl channels as well as nonselective stretch-activated cation channels in Ehrlich ascites tumor cells.J. Membrane Biol. 129:13–36
Cornet, M., Delpire, E., Gilles, R. 1987. Study of microfilaments network during volume process of cultured PC12 cells.Pfluegers Arch. 410:223–225
Cornet, M., Delpire, E., Gilles, R. 1988. Relations between cell volume control, microfilaments and microtubules networks in T2 and PC12 cultured cells.J. Physiol. (Paris) 83:43–49
Cornet M., Isobe, Y., Lemanski, L.F. 1992. Cytoskeleton of cultured PC12 cells during volume regulation under hypoosmotic conditions (submitted)
Cornet, M., Ubl, J., Kolb, H.-A. 1993. Cytoskeleton and ion movements during volume regulation in cultured PC 12 cells.J. Membrane Biol. (in press)
Delpire, E., Cornet, M., Gilles, R. 1991. Volume regulation in rat pheochromocytoma cultured cells submitted to hypoosmotic conditions.Arch. Int. Physiol. Biochim. 99:71–76
Delpire, E., Duchêne, C., Goessens, G., Gilles, R. 1985. Effects of osmotic shocks on the ultrastructure of different tissues and cell types.Exp. Cell Res. 160:106–116
De Mey, J., Moeremans, M., Geuens, G., Nuydens, R., de Brabander, M. 1981. High resolution light and electron microscopic localization of tubulin with the IgS (Immunogold Staining) method.Cell Biol. Int. Reports 5:889–899
Erxleben, C., Ubl, J., Kolb, H.-A. 1989. Identifying and characterizing stretch-activated channels.In: Molecular Neurobiology. A Practical Approach. H. Wheal, and J. Chad, editors. pp. 75–91. IRL, Oxford, Washington DC
Foskett, J.K., Spring, K.R. 1985. Involvement of calcium and cytoskeleton in gallbladder epithelial cell volume regulation.Am. J. Physiol. 248:C27-C36
Galkin, A.A., Khodorov, B.I. 1988. The involvement of furosemide-sensitive ion counter transport system in the autoregulation of macrophage volume: role of the cytoskeleton.Biol. Memb. 5(3):302–307
Gilles, R., Delpire, E., Duchêne, C., Cornet, M., Pequeux, A. 1986. The effect of cytochalasin B on the volume regulation response of isolated axons of the green crabCarcinus maenas submitted to hypo-osmotic media.Comp. Biochem. Physiol. 85A:523–525
Glenney, J.R., Jr., Bretscher, A., Weber, K. 1980. Calcium control of the intestinal microvillus cytoskeleton: its implication for the regulation of microfilament organization.Proc. Natl. Acad. Sci. USA 77(11):6458–6462
Grinstein, S., Du Pre, A., Rothstein, A. 1982. Volume regulation by human lymphocytes. Role of calcium.J. Gen. Physiol. 79:849–868
Hendil, K.B., Hoffmann, E.K. 1974. Cell volume regulation in Ehrlich ascites tumor cells.J. Cell. Physiol. 84:115–125
Henson, J.H., Schatten, G. 1983. Calcium regulation of the actinmediated cytoskeletal transformation of sea urchin coelomocytes.Cell Motil. 3:525–534
Hoffmann, E.K., Simonsen, L.O., Lambert, I.H. 1984. Volumeinduced increase of K+ and Cl− permeabilities in Ehrlich ascites tumor cells. Role of internal Ca2+.J. Membrane Biol. 78:211–222
Hoffmann, E.K., Lambert, I.H., Simonsen, L.O. 1986. Separate, Ca2+-activated K+ and Cl− transport pathways in Ehrlich ascites tumor cells.J. Membrane Biol. 91:227–244
Hoffmann, E.K., Lambert, I.H., Simonsen, L.O. 1988. Mechanisms in volume regulation in Ehrlich ascites tumor cells.Renal Physiol. Biochem. 11(3-5):221–247
Hoffmann, E.K., Simonsen, L.O., Lambert, I.H. 1993. Cell volume regulation: Intracellular Transmission.In: Advances in Comparative and Environmental Physiology. Vol 14, pp. 188–248. Springer-Verlag, Heidelberg.
Kleinzeller, A. 1965. The volume regulation in some animal cells.Arch. Biol. 76:217–232
Kleinzeller, A. 1972. Cellular transport of water.In: Metabolic Pathways. E. Hokin, editor. pp. 91–131. Academic, New York
Kramhøft, B., Lambert, I.H., Hoffmann, E.K., Jørgensen, F. 1986. Activation of Cl-dependent K transport in Ehrlich ascites tumor cells.Am. J. Physiol. 251:C369-C379
Lambert, I.H., Hoffmann, E.K., Jørgensen, F. 1989. Membrane potential, anion and cation conductance in Ehrlich ascites tumor cells.J. Membrane Biol. 111:113–132
Lewis, S.A., Moura, J.L.C. 1982. Incorporation of cytoplasmatic vesicles into apical membrane of mammalian urinary epithelium.Nature 298:685–688
Mac Lean-Fletcher, S.D., Pollard, T.D. 1980. Mechanism of action of cytochalasin B on actin.Cell 20:329–341
Mills, J.W. 1987. The cell cytoskeleton: possible role in volume control.Cur. Topics Memb. Transp. 30:75–101
Mills, J.W., Skiest, D.J. 1985. Role of cyclic AMP and cytoskeleton in volume control in MDCK cells.Mol. Physiol. 8:247–262
Mills, J.W., Lubin, M. 1986. Effect of adenosine 3′,5′-cyclic monophosphate on volume and cytoskeleton of MDCK cells.Am. J. Physiol. 250:C319-C324
Morris, C.E. 1990. Mechanosensitive ion channels.J. Membrane Biol. 113:93–107
Pardee, J.D., Simpson, P.A., Stryer, L., Spudich, J.A. 1982. Actin filaments undergo limited subunit exchange in physiological salt conditions.J. Cell Biol. 94:316–324
Pierce, S.K., Politis, A.D. 1990. Ca-activated cell volume recovery mechanisms.Annu. Rev. Physiol. 52:27–42
Pollard, T.D., Aebi, U., Copper, J.A., Elzinga, M., Fowler, W.E., Griffith, L.M., Herman, I.M., Heuser, J., Isenberg, G., Kiehart, D.P., Levy, J., Mac Lean-Fletcher, S.D., Maupin, P., Mooseker, M.S., Runge, M., Smith, P.R., Tseng, P. 1982. The mechanism of actin-filament assembly and crosslinking.In: Cell and Muscle Motility. R.M. Dowben, and J.W. Shay, editors. Vol. 2. pp. 15–44. Plenum, New York, London
Pritchard, K., Moody, C.J. 1986. Caldesmon: a calmodulin binding actin-regulatory protein.Cell Calcium 7(5-6):309–327
Rugolo, M., Mastrocola, T., Flamigni, A., Lenaz, G. 1989. Chloride transport in human fibroblasts is activated by hypotonic shock.Biochem. Biophys. Res. Comm. 160(3):1330–1338
Sachs, F. 1987. Baroreceptor mechanisms at the cellular level.Fed. Proc. 46(1):12–16
Sachs, F. 1988. Mechanical transduction in biological systems.CRC Crit. Rev. Biomed. Eng. 16:141–169
Sarkadi, B., Cheung, R., Mack, E., Grinstein, S., Gelfand, E.W., Rothstein, A. 1985. Cation and anion transport pathways in volume regulatory response of human lymphocytes to hyposmotic media.Am. J. Physiol. 248:C480-C487
Schliwa, M. 1986. The cytoskeleton. An introductory survey. Cell Biology Monographs. Vol. 13. Springer-Verlag, Wien, New York
Sobue, K., Sellers, J.R. 1991. Caldesmon, a novel regulatory protein in smooth muscle and nonmuscle actomyosin systems.J. Biol. Chem. 266:12115–12118
Stolz, B., Bereiter-Hahn, J. 1988. Increase of cytosolic calcium results in formation of F-actin aggregates in endothelial cells.Cell Biol. Int. Reports 12(4):321–329
Stossel, T.P. 1989. From signal to pseudopod.J. Biol. Chem. 264(31):18261–18264
Ubl, J., Murer, H., Kolb, H.-A. 1988. Ion channels activated by osmotic and mechanical stress in membranes of opossum kidney cells.J. Membrane Biol. 104:223–232
Ubl, J., Murer, H., Kolb, H.-A. 1989. Osmotically activated ion channels in membranes of opossum kidney cells.In: Salt and Hypertension. R. Rettig, D. Ganten, and F.C. Luft, editors. pp. 23–32. Springer-Verlag, Berlin, Heidelberg
Valdeolmillos, M., Garcia-Sancho, J., Herreros, B. 1982. Ca2+-dependent K+ transport in the Ehrlich ascites tumor cell.Biochim. Biophys. Acta 685:273–278
Van Rossum, G.D.V., Russo, M.A. 1981. Ouabain-resistant mechanism of volume control and the ultrastructural organization of liver slices recovering from swelling in vitro.J. Membrane Biol. 59:191–209
Wegner, A., Neuhaus, J.M. 1981. Requirement of divalent cations for fast exchange of actin monomers and actin filaments subunits.J. Mol. Biol. 153:681–693
Westermark, B., Porter, K.R. 1982. Hormonally induced changes in the cytoskeleton of human thyroid cells in culture.J. Cell Biol. 94:42–50
Wong, S., Chase, H.S., Jr. 1986. Role of intracellular calcium in cellular volume regulation.Am. J. Physiol. 250:C841-C852
Wulf, E., Deboben, A., Bautz, F.A., Faulstich, H. Wieland, T. 1979. Fluorescent phallotoxin, a tool for the visualization of cellular actin.Proc. Natl. Acad. Sci. USA 76(9):4498–4502
Yin, H.L., Hartwig, J.H., Maruyama, K., Stossel, T.P. 1981. Ca2+ control of actin filament length.J. Biol. Chem. 256:9693–9697
Ziyadeh, F.N., Mills, J.W., Kleinzeller, A. 1992. Hypotonicity and cell volume regulation in shark rectal gland: role of organic osmolytes and F-actin.Am. J. Physiol. 262:F468-F479.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Cornet, M., Lambert, I.H. & Hoffmann, E.K. Relation between cytoskeleton, hypo-osmotic treatment and volume regulation in Ehrlich ascites tumor cells. J. Membrain Biol. 131, 55–66 (1993). https://doi.org/10.1007/BF02258534
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02258534