Summary
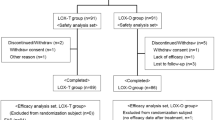
The efficacy and safety of local action transcutaneous flurbiprofen 40 mg [flurbiprofen LAT] patches and diclofenac sodium tablets, 50 mg b.d., were compared in an open, multicentre, randomized, parallel-group study in patients with soft-tissue rheumatism. Patches were replaced at 12-hourly intervals. Clinical assessments were performed after 7 and 14 days of treatment. Fifty-six patients were treated with flurbiprofen LAT and 53 with diclofenac. Six withdrawals (three from each group) occurred during the treatment period.
A statistically significant difference was observed in favour of flurbiprofen LAT for the principal measure, namely the investigator's opinion of overall change in clinical condition: 49/53 (92%) patients treated with flurbiprofen LAT had improved by day 14 compared with 36/49 (73%) patients receiving diclofenac sodium (p=0.03; eligible dataset). There were also statistically significant differences in favour of flurbiprofen LAT for the investigator's assessments of the overall severity of the clinical condition (p=0.03; eligible dataset), for the severity of pain at the region treated (p=0.04; intent-to-treat), and for the severity of tenderness (p<0.001; intent-to-treat). Supplementary analgesia (paracetamol) was required by two patients in the flurbiprofen LAT group and by eight diclofenac-treated patients. The difference in favour of flurbiprofen LAT group and by eight diclofenac-treated patients. The difference in favour of flurbiprofen LAT in the average daily consumption of paracetamol was significant (p=0.04). The patients' assessment of severity of pain on movement also favoured flurbiprofen LAT (p =0.049; eligible dataset), but there were no statistically significant differences in day or night pain or quality of sleep. For the patients' opinion of treatment there was, however, a statistically significant difference in favour of flurbiprofen LAT (p=0.02). Of the patients receiving flurbiprofen LAT, 94% regarded it as a convenient form of treatment.
With respect to tolerability 8/56 (14%) patients applying flurbiprofen patches reported a total of nine adverse effects (AEs) (mainly local, mild skin irritations), vs 9/52 (17%) patients receiving diclofenac, who reported 12 AEs. Most AEs in the enteric-coated diclofenac group were of a gastrointestinal nature (one of which was severe). In terms of the proportion of patients reporting AEs related to the digestive system, there was a statistically significant difference in favour of flurbiprofen LAT (p=0.011).
In conclusion, local treatment of soft-tissue rheumatism with flurbiprofen LAT was demonstrably superior to benchmark oral therapy with diclofenac sodium over a 2-week period in terms of both efficacy and gastrointestinal tolerability. Flurbiprofen LAT provided both an effective and convenient form of topical SAID treatment.
Similar content being viewed by others
References
Moser U, Waldburger M, Schwarz HA, Gobelet CA. A double-blind randomised multicentre study with tenoxicam, piroxicam and diclofenac sodium retard in the treatment of ambulant patients with osteoarthritis and extraarticular rheumatism. Schweiz Rundsch Med Prax 1989;78: 263–69.
Steele L, Hunneyball IM, Bresloff P. Comparison of rheumatoid synovial microsomes and bovine seminal- vesicle microsomes for determining the relative potencies of prostaglandin synthetase inhibitors. J Pharm Methods 1981;5:341–45.
Brodgen RN, Heel RC, Speight TM, Avery GS. Flurbiprofen: a review of its pharmacological properties and therapeutic use in rheumatic diseases. Drugs 1979;18:417–438.
Brooks CD, Linet OI, Schellenberg Turner LF et al. Clinical safety of flurbiprofen. J Clin Pharmacol 1990;30:342–51.
Watson Buchanan W, Kassam YB. European experience with flurbiprofen. A new analgesic/anti-inflammatory agent. Am J Med 1986;80:145–52.
Guy RH, Maibach HI. Drug delivery to local subcutaneous structure following topical administration. J Pharm Sci 1983;72:1375–80.
Wildfang IL, Maibach HI. Topical application of NSAIDs. In: Therapeutic applications of NSAIDs, Famaey JP, Paulus HE, editors. New York, Marcel Dekker, 1992.
Sugarawa S. A study of the concentration in the synovial fluid and tissues of patients treated with the transdermal topical application of flurbiprofen (FP-A). Ther Res 1987;6:298–94.
Frostick SP, Varley G, Smith C. The absorption of flurbiprofen from a topical preparation in patients undergoing arthroscopy. Clin Rheumatol 1994;13:356.
Taburet AM, Singlas E, Glass RC et al. Pharmacokinetic comparison of oral and local action transcutaneous flurbiprofen in healthy volunteers. J Clin Pharm Therapeutic 1995;20:101–7.
Poul J, West J, Buchanan N, Grahame R. Local action transcutaneous flurbiprofen in the treatment of soft-tissue rheumatism. Br J Rheumatol 1993;32:1000–3.
Aoki T, Sugarawa S, Fujimaki Y et al. Clinical evaluation of topical application of flurbiprofen (FP-A) as a treatment for periarthritis scapulohumeralis — collaborative double-blind test compared with the control. Jpn J Inflamm 1989;9:335–44.
Grahame R, Mattara L, Aoki K, Shichikawa K. A meta-analysis to compare flurbiprofen LAT to placebo in the treatment of soft-tissue rheumatism. Clin Rheumatol 1994;13:357.
Muldoon C, Earl R, Rees J. Safety and tolerability of flurbiprofen LAT. Clin Rheumatol 1994; 13:357.
Anonymous. Topical NSAIDs: a gimmick or a godsend? Lancet 1989;2:779–80.
Anonymous. More topical NSAIDs: worth the rub? Drug Ther Bull 1990;28:27–8.
Downie WW. Diclofenac/misoprostol. A review of the major clinical trials evaluating its efficacy and upper gastrointestinal tolerability in rheumatoid arthritis and osteoarthritis. Drugs 1993;45:1–6.
Zuinen C. Diclofenac/misoprostol vs diclofenac/placebo in treating acute episodes of tendinitis/bursitis of the shoulder. Drugs 1993;45:17–23.
Garza Elizondo MA, Mondragon VM. Flurbiprofen versus diclofenac in rheumatic disease of the musculoskeletal system. Invest Med Int 1990;17:16–22.
Atra E, Metz CA, Brown BL, Teoh K. Flurbiprofen versus diclofenac for the treatment of osteoarthritis of the knee. DICP 1990;24:920–23.
Jensen MP, Karoly P, Sanford B. The measurement of clinical pain intensity: a comparison of six methods. Pain 1986;27:117–26.
Winer BJ. Statistical principles in experimental design, 2nd ed., McGraw Hill, New York. 1971;752–811.
Armitage P, Berry G. Statistical methods in medical research, 2nd ed., Blackwell, Oxford. 1987;106–8.
SAS/STAT Users' guide vol. 1. ANOVA-FREQ, version 6. 4th ed., CARY NC.SAS Institute Inc. 1990.
SAS/STAT Users' guide vol. 2, GLM-VARCOMP, version 6. 4th ed., CARY NC. SAS Institute Inc. 1990.
Brownlee KA. Statistical theory and methodology in science and engineering, 2nd ed., Wiley, New York. 1965;482–89.
Koch GG, Edwards S. Clinical efficacy trials with categorical data. In: Biopharmaceutical statistics for drug development, Peace KE, editors. Marcel Dekker, New York. 1988;418–21.
Gould AL. A new approach to the analysis of clinical trial drugs with withdrawals. Biometrics 1980;36:721–27.
Hollander M, Wolfe DA. Nonparametric statistical methods, Wiley, New York. 1973;67–75.
Chlud K. Percutaneous therapy of painful arthritis. Ther Umsch 1991;48:42–5.
Bouchier-Hayes TA, Rotman H, Darekar BS. Comparison of the efficacy and tolerability of diclofenac gel (Voltarol Emugel) and felbinac gel (Traxam) in the treatment of soft tissue injuries. Br J Clin Pract 1990;44:19–20.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Martens, M. Efficacy and tolerability of a topical NSAID patch (local action transcutaneous flurbiprofen) and oral diclofenac in the treatment of soft-tissue rheumatism. Clin Rheumatol 16, 25–31 (1997). https://doi.org/10.1007/BF02238759
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02238759