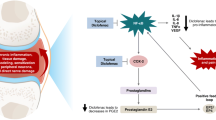
Abstract
Loxoprofen (Loxonin®, Loxonin® Pap, Loxonin® Tape) is a prodrug-type NSAID that is available in several formulations, including 60 mg tablets, 100 mg hydrogel patches and 50 or 100 mg tape. In active comparator-controlled trials, oral loxoprofen therapy (ranging from 2 days to 6 weeks’ duration depending on the pain type) provided analgesic efficacy that generally did not significantly differ from that of celecoxib for postoperative pain or frozen shoulder, ibuprofen for knee osteoarthritis or naproxen for lumbar pain. In double-blind, double-dummy, multicentre trials, loxoprofen hydrogel patches were noninferior to oral loxoprofen with regard to rates of final overall symptomatic improvement over 1–4 weeks in patients with knee osteoarthritis, myalgia or trauma-induced swelling and pain. Loxoprofen hydrogel patches were also noninferior to other commercially available patches (ketoprofen and indometacin) over 2 or 4 weeks in patients with knee osteoarthritis or myalgia in open-label studies. Oral and topical loxoprofen were generally well tolerated in clinical trials. Thus, loxoprofen is a useful analgesic option for patients with pain and inflammation, with topical loxoprofen potentially reducing the risk of gastrointestinal, cardiovascular and renal complications associated with oral NSAID use.
Similar content being viewed by others
References
Burmester G, Lanas A, Biasucci L, et al. The appropriate use of non-steroidal anti-inflammatory drugs in rheumatic disease: opinions of a multidisciplinary European expert panel. Ann Rheum Dis. 2011;70(5):818–22.
Hochberg MC, Altman RD, April KT, et al. American College of Rheumatology 2012 recommendations for the use of nonpharmacologic and pharmacologic therapies in osteoarthritis of the hand, hip, and knee. Arthritis Care Res. 2012;64(4):465–74.
Arakawa T, Fujiwara Y, Sollano JD, et al. A questionnaire-based survey on the prescription of non-steroidal anti-inflammatory drugs by physicians in East Asian countries in 2007. Digestion. 2009;79(3):177–85.
Moore N, Pollack C, Butkerait P. Adverse drug reactions and drug-drug interactions with over-the-counter NSAIDs. Ther Clin Risk Manag. 2015;11:1061–75.
Scarpignato C, Lanas A, Blandizzi C, et al. Safe prescribing of non-steroidal anti-inflammatory drugs in patients with osteoarthritis—an expert consensus addressing benefits as well as gastrointestinal and cardiovascular risks. BMC Med. 2015;13:[article no. 55].
Scheiman JM. NSAID-induced gastrointestinal injury: a focused update for clinicians. J Clin Gastroenterol. 2016;50(1):5–10.
Antman EM, Bennett JS, Daugherty A, et al. Use of nonsteroidal antiinflammatory drugs: an update for clinicians: a scientific statement from the American Heart Association. Circulation. 2007;115(12):1634–42.
Rannou F, Pelletier JP, Martel-Pelletier J. Efficacy and safety of topical NSAIDs in the management of osteoarthritis: evidence from real-life setting trials and surveys. Semin Arthritis Rheum. 2016;45(4 Suppl):S18–21.
Bruyère O, Cooper C, Pelletier JP, et al. A consensus statement on the European Society for Clinical and Economic Aspects of Osteoporosis and Osteoarthritis (ESCEO) algorithm for the management of knee osteoarthritis—from evidence-based medicine to the real-life setting. Semin Arthritis Rheum. 2016;45(4 Suppl):S3–11.
Daiichi Sankyo Co Ltd. Loxonin® Pap 100 mg, Tape 50 mg and 100 mg (loxoprofen sodium hydrate patches): Japanese interview form on drugs. 2015. http://www.pmda.go.jp. Accessed 8 Jan 2016.
Daiichi Sankyo Co Ltd. Loxoprofen sodium tablets: Chinese prescribing information. 2009. http://www.daiichisankyo.com.cn. Accessed 2 Mar 2016.
Daiichi Sankyo Co Ltd. Loxoprofen patches: Chinese prescribing information. 2015. http://www.daiichisankyo.com.cn. Accessed 2 Mar 2016.
Daiichi Sankyo Co Ltd. Loxonin® Pap 100 mg, Tape 50 mg and 100 mg, Gel 1 %: Japanese summarized product information. 2014.
Tanaka K, Terada A, Iizuka Y, et al. Loxoprofen sodium (CS-600), a new non-steroidal anti-inflammatory drug. Ann Rep Sankyo Res Lab. 1984;36:1–43.
Noguchi M, Kimoto A, Gierse JK, et al. Enzymologic and pharmacologic profile of loxoprofen sodium and its metabolites. Biol Pharm Bull. 2005;28(11):2075–9.
Hamamoto T, Takeuchi S, Sasakura M, et al. Anti-inflammatory and analgesic effects of hydrogel patches containing loxoprofen sodium [Japanese with English abstract]. J Clin Ther Med. 2006;22(3):179–86.
Araki H, Kuwagata S, Soumura M, et al. Safety and efficacy of skin patches containing loxoprofen sodium in diabetic patients with overt nephropathy. Clin Exp Nephrol. 2014;18(3):487–91.
Kawano S, Tsuji S, Hayashi N, et al. Effects of loxoprofen sodium, a newly synthesized non-steroidal anti-inflammatory drug, and indomethacin on gastric mucosal haemodynamics in the human. J Gastroenterol Hepatol. 1995;10(1):81–5.
Mizukami K, Murakami K, Yamauchi M, et al. Evaluation of selective cyclooxygenase-2 inhibitor-induced small bowel injury: randomized cross-over study compared with loxoprofen in healthy subjects. Dig Endosc. 2013;25(3):288–94.
Sakamoto C, Kawai T, Nakamura S, et al. Comparison of gastroduodenal ulcer incidence in healthy Japanese subjects taking celecoxib or loxoprofen evaluated by endoscopy: a placebo-controlled, double-blind 2-week study. Aliment Pharmacol Ther. 2013;37(3):346–54.
Fujimori S, Hanada R, Hayashida M, et al. Celecoxib monotherapy maintained small intestinal mucosa better compared with loxoprofen plus lansoprazole treatment: a double-blind, randomized, controlled trial. J Clin Gastroenterol. 2016;50(3):218–26.
Daiichi Sankyo Co Ltd. Loxonin® (loxoprofen sodium) tablets 60 mg, fine granules 10 %: Japanese interview form on drugs. 2014. http://www.info.pmda.go.jp. Accessed 18 May 2016.
Sugawara S, Hasegawa S, Naganuma H, et al. Pharmacokinetic study of the hydrogel patch containing loxoprofen sodium (LX-A) following 5 days applications repeated once or twice a day [Japanese with English abstract]. J Clin Ther Med. 2006;22(4):279–92.
Chen X, Zhao Q, Hitsu E, et al. Dermatopharmacokinetic bioequivalence study of two types of topical patches containing loxoprofen sodium. Int J Clin Pharmacol Ther. 2014;52(10):927–32.
Chen X, Matsuzawa T, Hitsu E, et al. Model of the dermatopharmacokinetic profile of two loxoprofen patches for bioequivalence confirmation. Int J Clin Pharmacol Ther. 2015;53(5):412–3.
Matsuzawa T, Sairo H, Kurihara A, et al. Absorption, distribution, metabolism and excretion after dermal application of hydrogel patch containing loxoprofen sodium in rats [Japanese with English abstract]. J Clin Ther Med. 2006;22(3):187–203.
Onda A, Ogoshi A, Itoh M, et al. Comparison of the effects of treatment with celecoxib, loxoprofen, and acetaminophen on postoperative acute pain after arthroscopic knee surgery: a randomized, parallel-group trial. J Orthop Sci. 2016;21(2):172–7.
Sekiguchi H, Inoue G, Nakazawa T, et al. Loxoprofen sodium and celecoxib for postoperative pain in patients after spinal surgery: a randomized comparative study. J Orthop Sci. 2015;20(4):617–23.
Shi Y-Q, Han X-H. Clinical observation of loxoprofen sodium in treatment of patients with knee osteoarthritis [Chinese with English abstract]. Pharm Care Res. 2004;4(1):46–8.
Waikakul S, Soparat K. Effectiveness and safety of loxoprofen compared with naproxen in nonsurgical low back pain: a parallel study. Clin Drug Investig. 1995;10(1):59–63.
Ohta S, Komai O, Hanakawa H. Comparative study of the clinical efficacy of the selective cyclooxygenase-2 inhibitor celecoxib compared with loxoprofen in patients with frozen shoulder. Mod Rheumatol. 2014;24(1):144–9.
Aoki T. Effectiveness and safety of loxoprofen in elderly patients with lumbar pain. Drug Invest. 1992;4(6):477–83.
Sugawara S, Tateishi A, Tanaka M, et al. A dose finding study of a hydrogel patch containing loxoprofen sodium (LX-A) in patients with osteoarthritis of the knee (late phase II clinical study): a randomized, double-blind, placebo controlled comparative study [Japanese with English abstract]. J Clin Ther Med. 2006;22(4):293–310.
Sugawara S, Tateishi A, Tanaka M, et al. Evaluation of administration frequency of LX-A in patients with osteoarthritis of the knee: randomized, open label study compared between once-daily administration and twice-daily administration [Japanese with English abstract]. J Clin Ther Med. 2006;22(4):311–26.
Mu R, Bao CD, Chen ZW, et al. Efficacy and safety of loxoprofen hydrogel patch versus loxoprofen tablet in patients with knee osteoarthritis: a randomized controlled non-inferiority trial. Clin Rheumatol. 2016;35(1):165–73.
Zhao DB, Shi YQ, Li ZG, et al. Effectiveness and safety of hydrogel patches containing loxoprofen sodium in patients with myalgia: a randomized, controlled, double-blind, double-parallel, multicenter phase 3 trial [abstract no. APLAR-0134]. Int J Rheum Dis. 2013;16(Suppl 1):111.
Zhang H, Lin J, Sun T, et al. Double-blind, multicenter trial to evaluate safety and efficacy of hydrogel patch containing loxoprofen-sodium in treating swelling and pain caused by trauma [abstract no. APLAR-0153]. Int J Rheum Dis. 2013;16(Suppl 1):111.
Sugawara S, Kuroki Y, Tateishi A, et al. The effects of Loxonin® Pap 100 mg (hydrogel patch containing loxoprofen sodium) for the treatment of patients with osteoarthritis of the knee: randomized, double-blind clinical study with loxoprofen sodium tablets [Japanese with English abstract]. J Clin Ther Med. 2006;22(5):393–409.
Sugawara S, Kuroki Y, Tanaka M, et al. The effects of Loxonin® Pap 100 mg (hydrogel patch containing loxoprofen sodium) for the treatment of myalgia: randomized, double-blind study compared with loxoprofen sodium tablets [Japanese with English abstract]. J Clin Ther Med. 2006;22(5):411–26.
Sugawara S, Iwasaki Y, Aoki T. The effects of Loxonin® Pap 100 mg (hydrogel patch containing loxoprofen sodium) for the treatment of swelling or pain following injuries: randomized, double-blind study with loxoprofen sodium tablets [Japanese with English abstract]. J Clin Ther Med. 2006;22(5):427–42.
Sugawara S, Nobuoka F, Ogawa D, et al. Clinical efficacy of the hydrogel patch containing loxoprofen sodium (LX-A) on osteoarthritis of the knee: a randomized, open label clinical study with ketoprofen patch (phase III therapeutic confirmatory study) [Japanese with English abstract]. J Clin Ther Med. 2007;23(1):55–71.
Sugawara S, Nobuoka F, Ogawa D, et al. Clinical efficacy of the hydrogel patch containing loxoprofen sodium (LX-A) on myalgia: a randomized, open label clinical study compared with indometacin patch (phase III therapeutic confirmatory study) [Japanese with English abstract]. J Clin Ther Med. 2007;23(2):127–41.
Mizutani H, Miyano F, Uozu A, et al. Drug use result survey of Loxonin® Tape (patch containing loxoprofen sodium hydrate) [Japanese with English abstract]. J Clin Ther Med. 2010;26(10):727–41.
Mizutani H, Miyano F, Uozu A, et al. Drug use result survey of Loxonin® Pap (hydrogel patch containing loxoprofen sodium hydrate) [Japanese with English abstract]. J Clin Ther Med. 2010;26(3):227–40.
Chinese Orthopaedic Association. Diagnosis and treatment of osteoarthritis. Orthop Surg. 2010;2(1):1–6.
Sakamoto C, Soen S. Efficacy and safety of the selective cyclooxygenase-2 inhibitor celecoxib in the treatment of rheumatoid arthritis and osteoarthritis in Japan. Digestion. 2011;83(1–2):108–23.
Acknowledgments
During the peer review process, the manufacturer of loxoprofen was also offered an opportunity to review this article. Changes resulting from comments received were made on the basis of scientific and editorial merit.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
The preparation of this review was not supported by any external funding.
Conflict of interest
Sarah Greig and Karly Garnock-Jones are salaried employees of Adis/Springer, are responsible for the article content and declare no relevant conflicts of interest.
Additional information
The manuscript was reviewed by: X. Chen, Clinical Pharmacology Research Center, Peking Union Medical College Hospital, Beijing, China; H.-G. Xie, General Clinical Research Center, Nanjing First Hospital, Nanjing Medical University, Nanjing, China.
Rights and permissions
About this article
Cite this article
Greig, S.L., Garnock-Jones, K.P. Loxoprofen: A Review in Pain and Inflammation. Clin Drug Investig 36, 771–781 (2016). https://doi.org/10.1007/s40261-016-0440-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40261-016-0440-9