Summary
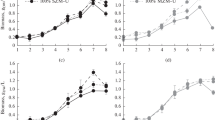
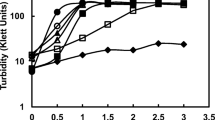
The optimum conditions for growth ofFrankia sp. HFPCcI3 isolated fromCasuarina cunninghamiana, were studied in batch culture using defined media. Maximum growth (doubling time was 24 h)_was achieved at 33°C and at pH 6.3 with pyruvate and NH +4 as the sole C and N sources, respectively. Removal of NH +4 from the culture medium resulted in vesicle differentiation which was paralleled by induction of acetylene reduction activity. Growth on atmospheric N2 was optimal with combined pyruvate and glucose as the carbon sources and displayed a doubling time of about 48 h. Comparisons in growth and N2-fixing activity ofFrankia strains grown in a variety of cultural conditions demonstrate the range of behavior among the strains.
Similar content being viewed by others
References
Akkermans A D L, Roelofsen W, Blom, J, Huss-Danell K and Harkink R 1983 Utilization of carbon and nitrogen compounds by Frankia in synthetic media and in root nodules ofAlnus glutinosa, Hippophae rhamnoides andDatisca cannabina. Can. J. Bot. 61, 2793–2800.
Blom J 1981 Utilization of fatty acids and NH +4 by Frankia AvcI1. FEMS Microbiol. Letters 10, 143–145.
Blom J 1982 Carbon and nitrogen source requirements ofFrankia strains. FEMS Microbiol. Letters 13, 51–55.
Blom J and Harkink R 1981 Metabolic pathways for gluconeogenesis and energy generation inFrankia AvcI1. FEMS Microbiol. Letters 11, 221–224.
Blom J, Roelofsen W and Akkermans A D L 1980 Growth ofFrankia AvcI1 on media containing Tween 80 as C-source. FEMS Microbiol. Letters 9, 131–135.
Bond G and Mackintosh A H 1975 Diurnal changes in nitrogen fixation in the root nodules of Casuarina. Proc. Roy. Soc. Lond. (Ser. B). 192 1–12.
Bradford M M 1976 A rapid and sensitive method for quantification of microgram quantities of protein utilizing the principle of protein dye binding. Anal. Biochem. 72, 248–254.
Burggraaf A J P and Shipton W A 1982 Estimates ofFrankia growth under various pH and temperature regimes. Plant and Soil 69, 135–147.
Burggraaf A J P and Shipton W A 1983 Studies on the growth of Frankia isolates in relation to infectivity and nitrogen fixation (acetylene reduction). Can. J. Bot. 61, 2774–2782.
Burggraaf A J P and Valstar J 1984 Heterogeneity withinFrankia sp. LDAgpl studied among clones and reisolates. Plant and Soil 78, 29–43.
Callaham D, Del Tredici P and Torrey J G 1978 Isolation and cultivationin vitro of the actinomycete causing root nodulation in Comptonia. Science 199, 899–902.
Drews G 1965 Untersuchungen zur Regulation der Bacteriochlorophyll-Synthese beiRhodospirillum rubrum. Arch. Mikrobiol. 51, 186–198.
Fontaine M S, Lancelle S A and Torrey J G 1984 Initiation and ontogeny of vesicles in culturedFrankia strain HFPArI3. J. Bact. 160, 921–927.
Lechevalier M P 1964 The taxonomy of the genusFrankia. Plant and Soil 78, 1–6.
Lechevalier, M P, Baker D and Horriere F 1983 Physiology, chemistry, serology and infectivity of twoFrankia isolates fromAlnus incana subsp.rugosa. Can. J. Bot. 61, 2826–2833.
Lechevalier M P and Lechevalier H A 1979 The taxonomic position of the actinomycetic endophytes. pp. 111–122.In Symbiotic Nitrogen Fixation in the Management of Temperate Forests. Eds. J C Gordon, C T Wheeler and D A Perry Forest Research Laboratory, Oregon State Univ., Corvallis, Oregon.
Lechevalier M P and Ruan J-S 1984 Physiology and chemical diversity ofFrankia spp. isolated from nodules ofComptonia peregrina (L.) Coult. andCeanothus americanus L. Plant and Soil 78, 15–22.
Lopez M F, Fontaine M S and Torrey J G 1984 Levels of trehalose and glycogen inFrankia sp. HFPArI3 (Actinomycetales). Can. J. Microbiol. 30, 746–752.
Murry M A, Fontaine M S and Torrey J G 1984 Growth kinetics and nitrogenase induction inFrankia sp. HFPArI3 grown in batch culture. Plant and Soil 78, 61–78.
Shipton W A and Burggraaf A J P 1982 A comparison of the requirements for various carbon and nitrogen sources and vitamins in someFrankia isolates. Plant and Soil 69, 149–161.
Shipton W A and Burggraaf A J P 1983 Aspects of the cultural behaviour ofFrankia and possible ecological implications. Can. J. Bot. 61, 2783–2792.
Tisa L, McBride M and Ensign J C 1983 Studies of growth and morphology ofFrankia strains EAN1pec, EuI1c, CpI1, and ACN1AG. Can. J. Bot. 61, 1768–2773.
Tjepkema J, Ormerod W and Torrey J G 1980 Vesicle formation and acetylene reduction activity inFrankia sp. CpI1 cultured in defined nutrient media. Nature 287, 633–635.
Tjepkema J, Ormerod W and Torrey J G 1981 Factors affecting vesicle formation and acetylene reduction (nitrogenase activity) inFrankia sp. CpI1. Can. J. Microbiol. 27, 915–823.
Wilcockson J and Werner D 1979 Organic acids, and prolonged nitrogenase activity of non-growing free-livingRhizobium japonicum Arch. Microbiol. 122, 153–159.
Zhang Z, Lopez M F and Torrey J G 1984 A comparison of cultural characteristics and infectivity ofFrankia isolates from root nodules ofCasuarina species. Plant and Soil 78, 78–90.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zhongze, Z., Murry, M.A. & Torrey, J.G. Culture conditions influencing growth and nitrogen fixation inFrankia sp.HFPCcI3 isolated from Casuarina. Plant Soil 91, 3–15 (1986). https://doi.org/10.1007/BF02181814
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02181814