Abstract
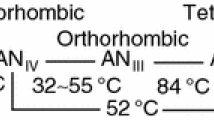
Ammonium uranate was precipitated from uranyl nitrate solution using gaseous ammonia, then filtered, washed with demineralized water and dried. The influence of pH and ammonia flow rate on their composition and structure were examined by X-ray diffraction analysis, making use of additional information obtained from infrared analysis.
Similar content being viewed by others
References
S.A. Rodríguez, Ch.A.L. Acosta, L.A. Fucugauchi, B.L.R. Macías,J. Radioanal. Nucl. Chem., Lett., 175 (1993) 63.
K. Nakamoto, Infrared Spectra of Inorganic and Coordination Compounds, Wiley, New York, N.Y., 1963.
R.A. Nyquist, R.O. Kagel, Infrared Spectra of Inorganic Compounds, Academic Press, New York, N.Y., 1971.
I. Ahuja, G.L. Yadava, S. Tripathi,Indian Journal of Chemistry, 28A (1989) 167.
L. Chen-Yeh, Y. Yu-Chay, P.R. Griffiths,J. Radioanal. Nucl. Chem., Articles, 131 (1989) 131.
A.J. Paul, P.M.A. Sherwood,Appl. Spectroscopy, 40 (1986) 519.
V. Urbanek, V. Sara, J. Moravec,J. Inorg. Nucl. Chem., 41 (1979) 537.
W.I. Stuart,J. Inorg. Nucl. Chem., 38 (1976) 1378.
S.A. Rodríguez, B.E.M. López, L.A. Fucugauchi,Lanthanide and Actinide Research, 3 (1989) 105.
S.A. Rodríguez, B.E.M. López, L.A. Fucugauchi,Rev. Soc. Quim. Mex., 35 (1991) 64.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rodríguez, A.S., Macías, L.R.B. Effect of pH and gaseous ammonia flow rate on the composition and structure of ammonium uranate. Journal of Radioanalytical and Nuclear Chemistry Letters 186, 205–212 (1994). https://doi.org/10.1007/BF02162751
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02162751