Summary
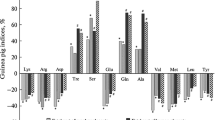
Two membrane associated placental tissue proteins (PP4 and MP1) have been isolated and characterized. Both proteins are found in the soluble as well as solubilized protein fractions of the human placenta and thus appear to be at least partly associated with placental membranes. PP4 has a molecular weight of 35000 and apparently consists of a single peptide chain. It has an electrophoretic mobility in between the α1- and α2-globulins, an isoelectric point of 4.85 and a sedimentation coefficient of 3.3 S. The carbohydrate content of PP4 amounts to 2.4%. MP1 was isolated from placental protein fractions solubilized with Triton X-100. It has a molecular weight of around 180000 and appears to be composed of two identical subunits which are non-covalently linked. MP1 was found to have an electrophoretic mobility in between the α2- and β1-globulins, an isoelectric point of 4.75 and a sedimentation coefficient of 6.65 S. MP1 is a glycoprotein which contains 9.6% carbohydrates. Immunochemical methods were used to detect and quantitate PP4 and MP1 in extracts of placentae and other human tissues. MP1 appears to be specific to the placenta, whereas PP4 was found to occur also in certain other human tissues. The diagnostic significance of detection and measurement of these proteins in tissues and body fluids is presently under investigation.
Similar content being viewed by others
References
Axen R, Porath J, Ernback S (1967) Chemical coupling of peptides and proteins to polysaccharides by means of cyanogen halides. Nature 214: 1302–1304
Bohn H (1971) Nachweis und Charakterisierung von Schwangerschaftsproteinen in der menschlichen Plazenta sowie ihre quantitative immunologische Bestimmung im Serum schwangerer Frauen. Arch Gynaekol 210: 440–457
Bohn H (1979) Placental und pregnancy proteins. In: Lehmann FG (ed) Carcino-embryonic Proteins. Chemistry, Biology, Clinical Application, Vol I. Elsevier/North Holland. Biomedical Press, Amsterdam, pp 289–299
Bohn H (1982) New soluble placental tissue proteins (Characterization and possible diagnostic significance). Ric Clin Lab 12: 221–230
Bohn H (1983) Systematic identification of specific oncoplacental proteins. In: Fishman WH (ed) Oncodevelopmental markers. Academic Press, New York, pp 69–86
Bohn H, Kraus W, Winckler W (1983) Purification and characterization of two new soluble placental tissue proteins (PP13 and PP17). Oncodev Biol Med 4: 343–350
Duance VC, Bailey A (1983) Structure of the trophoblast basement membrane. In: Loke YW, Whyte A (eds) Biology of trophoblast. Elsevier Science Publishers, Amsterdam, pp 597–625
Inaba N, Sato N, Ijitchi M, Nito A, Sekiya S, Takamizawa H, Lüben G, Bohn H (1983) Immunocytochemical location of membrane-associated proteins, MP1 and PP4 in placental tissues, blood cells and tumor tissues. Oncodev Biol Med Abstract volumn (in press)
Wild AE (1983) Trophoblast cell surface receptors. In: Loke YW, Whyte A (eds) Biology of trophoblast. Elsevier Science Publishers, Amsterdam, pp 471–512
Zwisler O, Biel H (1966) Elektrophorese in horizontalem Polyacrylamidgel. Z Klin Chem 4: 58–60
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bohn, H., Kraus, W. & Winckler, W. Isolation and characterization of two membrane-associated placental tissue proteins. Arch. Gynecol. 236, 225–233 (1985). https://doi.org/10.1007/BF02133940
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02133940