Abstract
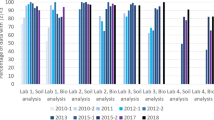
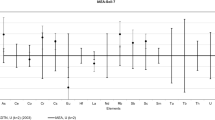
Increasingly govemmental bodies and industry require that supporting analytical laboratories have their quality assurance program implemented in a quality system by international standards such as derived from the ISO-25 guide. Neutron activation analysis (NAA) laboratories may have to deal with this trend too. In universities and research laboratories the need for it, and the implications of total quality management system are sometimes misconceived by unfamiliarity with the issue. The laboratory for INAA in Delft has been accredited for its quality system since 1993. Some of the tangible improvements since the introduction of quality management are presented. Four strategical considerations are given to consider the introduction of quality management at NAA laboratories, viz. with respect to the role of NAA for the validation of other methods, the role of NAA in the certification of reference materials, the preservation of knowledge and the acceptance of NAA as a respectable method.
Similar content being viewed by others
References
M. Thompson, R. Wood, Hamonised Guidelines for Intemal Quality Control in Analytical Chemistry Laboratories, IUPAC Technical Report, 1995.
P. Bode, J. P. J. van Dalen, J. Radioanal. Nucl. Chem., 179 (1994) 141.
P. Bode, Analyst, 120 (1995) 1527.
R. Dams, Pure Appl. Chem., 55 (1983) 1957.
R. Parr, in: Trace Element Analytical Chemistry in Medicine and Biology,P. Brätier, P. Schramel (Eds), De Gruyter, Berlin, 1980, p. 633.
D. A. Becker, Trans. Am. Nucl. Soc., 71 (1994) 24.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bode, P. From misconception to a must: The measured merits of total quality management and accreditation in INAA. J Radioanal Nucl Chem 215, 51–57 (1997). https://doi.org/10.1007/BF02109877
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02109877