Abstract
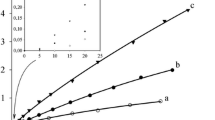
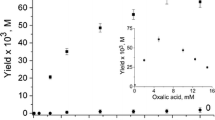
Selective oxidation of cyclohexane to cyclohexanone was carried out in a RuCl3-pyridine-acetic acid-KHSO5 mixture. This system yields 2.8% of cyclohexanone (based on ‘oxone’ concentration) under the conditions employed. Mechanistic postulate for the oxidation reaction involves the formation of a carbene intermediate (Ru=C) prior to the ketone formation.
Similar content being viewed by others
References
D. H. R. Barton, M. J. Gastiger, W. B. Motherwell: J. Chem. Soc. Chem. Commun., 41 (1983).
D. H. R. Barton, F. Halley, N. Ozbalik, M. Schinitt, E. Young, G. Balavoine: J. Am. Chem. Soc.,111, 7144 (1989).
G. Balavoine, D. H. R. Barton, J. Boivin, A. Gref, N. Ozbalik, H. Rivière: J. Chem. Soc. Chem. Commun., 1727 (1986).
D. H. R. Barton, F. Halley, N. Ozbalik, E. Young, G. Balavoine, A. Gref, J. Boivin: New J. Chem.,13, 177 (1989).
D. H. R. Barton, D. Doller, Y. V. Geletii: Mendeleev Commun., 115 (1991).
Y. Sasaki, M. Suzuki, A. Tobiwa, M. Ebihara, T. Yamaguchi, C. Katinlo, T. Ito: J. Am. Chem. Soc.,110, 6251 (1988).
M. M. Taqui Khan, D. Chatterjee, R. R. Merchant: J. Mol. Catal.,66, 23 (1991).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Taqui Khan, M.M., Chatterjee, D. & Bajaj, H.C. Selective oxidation of cyclohexane by a Ru(III) analogue of the gif-system. RuIIIacetic acid-pyridine-KHSO5 . React Kinet Catal Lett 49, 81–85 (1993). https://doi.org/10.1007/BF02084032
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02084032