Abstract
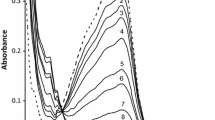
The kinetics of the title reaction was investigated spectrophotometrically in aqueous perchloric acid medium under isolated conditions, ([NH2OH]≫[Fe(III)]). The reaction was found to be first order in iron(III) as well as in the substrate. INcrease in the [H+] ion concentration of the medium were found to decrease the rate of the reaction, while ionic strength has little influence. Kinetic evidence for the formation of a 1∶2 complex between iron(III) and 1,10-phenanthroline has been obtained. Nitrogen was found to be the principal product of oxidation. A plausible mechanism consistent with the observed
Abstract
Кинетика заглавной реакции была исследована спектрофотометрически в среде водной хлорной кислоты и в условиях [NH2OH]≫[Fe(III)]. Реакция имеет первый порядок как по железу(III), так и по субстрату. Увеличение [H+] бдиэлектрической постоянной среды уменьшает скорость реакции, в то время как ионная сила едва оказывает какое-либо влияние. Были получены кинетические доказательства существования комплеса между жепезом(III) и 1,10-фенантролином с составом 1∶2. Азот был основным продуктом окиления. Предлагается вероятный механизм протекания процесса.
Similar content being viewed by others
References
K. Bhaskara Rao, G. Gopala Rao: Z. Anal. Chem.,157, 100 (1957).
G. Bengtsson: Acta. Chim. Scand.,27, 1717 (1973).
M. S. Frank, A. Kodanda Ramaiah, P. V. Krishna Rao: Indian J. Chem.,18(A), 369 (1979).
P. V. Subba Rao, B. A. N. Murthy, R. V. S. Murthy: Z. Phys. Chem. Leipzig,257, 4 (1979).
P. V. Subba Rao, B. A. N. Murthy, R. V. S. Murthy, K. S. Murthy: J. Indian. Chem. Soc.,LV, 207 (1978).
P. V. Krishna Rao, M. S. Frank: Indian J. Chem.,19(A), 538 (1980).
Abdul Fattah M. Nazer, Cecil F. Wells: J. Chem. Soc. Dalton, 1532 (1980).
I. M. Kolthoff, Belcher: ‘Volumetric Analysis’ Vol. 3. p. 525, Interscience Publications, New York, 1957.
W. A. Waters, I. R. Wilson: J. Chem. Soc., 534 (1966).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Vittal, A.S.P., Rao, P.V.K. & Rao, K.J.M. Kinetics of oxidation of hydroxylamine by iron(III) in presence of 1,10-phenanthroline. React Kinet Catal Lett 23, 175–179 (1983). https://doi.org/10.1007/BF02065685
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF02065685