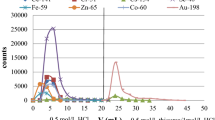
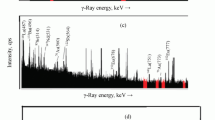
Abstract
Phosphorus at trace levels alters the properties of metals and alloys. Its determination was investigated by radiochemical neutron activation analysis. Separation by solvent extraction or by evolution as P0 showed to be neither selective nor quantitative in presence of a metallic matrix. Therefore, a new method of separation by reduction to phosphorus hydride followed by liquid scintillation counting was investigated. This method is quantitative in the case of non-radioactive iron doped with radioactive phosphorus. At present, the separation is not quantitative for irradiated iron samples, owing probably to hot atom or radiation effects. A detection limit of 0.002 μg is expected.
Similar content being viewed by others
References
Guide to Techniques and Applications of Atomic Spectrometry, Perkin-Elmer, 1988.
G. GOETHALS, C. VAN DE CASTEELE, J. HOSTE, Anal. Chim. Acta, 101 (1978) 63.
H. JASKOLSKA, L. ROWINSKA, J. Radioanal. Chem., 26 (1975) 31.
H. H. ROSS, R. B. HAHN, Talanta, 8, (1961) 575.
C. LOOS-NESKOVIC, M. FEDOROFF, J. Radioanal. Chem., 80 (1983) 97.
M. SHINOGI, I. MORI, Yakugaku Zasshi, 96 (1976) 1282.
J. E. DAVIES, R. GRZESKOWIAK, J. MENDMAN, J. Chromatography, 201 (1980) 305.
B. WELZ, M. MELCHER, Anal. Chem., 57 (1985) 427.
Z. VECERA, B. BICHER, Hutnicke Listy, 14 (1978) 56.
M. FEDOROFF, C. LOOS-NESKOVIC, V. N. SAMOSYUK, B. A. CHAPYZHNIKOV, J. Radioanal. Chem., 72 (1982) 715.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rouchaud, J.C., Fedoroff, M. Determination of phosphorus in metals by neutron activation and chemical separation as hydride. Journal of Radioanalytical and Nuclear Chemistry, Articles 174, 57–63 (1993). https://doi.org/10.1007/BF02040333
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02040333