Abstract
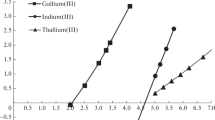
The toxicity of elements depends strongly on their chemical form. The separation of individual species must usually precede the determination by activation analysis or by other methods. The simple and rapid methods for the separation of inorganic mercury(II), methylmercury and phenylmercury compounds, arsenic(III) and arsenic(V) species including the separation of methylated forms, thallium(I) and thallium(III), chromium(III) and chromium(VI) using liquidliquid distribution reactions are presented.
Similar content being viewed by others
References
L. G. SILLÉN, A. E. MARTELL, Stability Constants of Metal Ion Complexes, The Chemical Society, London, 1964, Supplement No. 1, The Chemical Society, London, 1971.
J. STARÝ, H. FREISER, Equilibrium Constants of Liquid-Liquid Distribution Reactions, Part IV. Chelating Extractants. Pergamon Press, Oxford, 1978.
J. STARÝ, K. KRATZER, Radiochem. Radioanal. Letters, 28 (1977) 53.
J. STARÝ, K. KRATZER, J. PRÁŠILOVÁ, Anal. Chim. Acta, 100 (1978) 627.
J. STARÝ, J. PRÁŠILOVÁ, Radiochem. Radioanal. Letters, 26 (1976) 33.
J. STARÝ, J. PRÁŠILOVÁ, Radiochem. Radioanal. Letters, 26 (1976) 193.
J. STARÝ, A. ZEMAN, K. KRATZER, Radiochem. Radioanal. Letters, 52 (1982) 263.
J. STARÝ, B. HAVLÍK, J. PRÁSILOVÁ, K. KRATZER, J. HANUŠOVÁ, J. Environ, Anal. Chem., 5 (1978) 89.
T. SEXINE, Y. HASEGAWA, Solvent Extration Chemistry, M. Dekker, New York, 1977.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Starý, J. Rapid separation of mercury, arsenic, thallium and chromium species. Journal of Radioanalytical and Nuclear Chemistry, Articles 112, 119–123 (1987). https://doi.org/10.1007/BF02037282
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02037282