Abstract
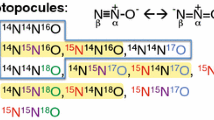
The rate of nitrogen isotope exchange between NO and HNO3 has been measured as a function of nitric acid concentration of 1.5–4M·1−1. The exchange rate law is shown to beR=k[HNO3]2[N2O3] and the measured activation energy isE=67.78kJ ·M−1 (16.2 kcal·M−1). It is concluded that N2O3 participates in15N/14N exchange between NO and HNO3 at nitric acid concentrations higher than 1.5M·1−1.
Similar content being viewed by others
References
W. SINDEL, T. I. TAYLOR, J. Chem. Phys., 33 (1955) 981.
W. SPINDEL, T. I. TAYLOR, Trans. N. Y. Acad. Sci. Ser. I., 19 (1958) 3.
T. I. TAYLOR, W. SPINDEL, Proc. Symp. on Istope Separation, Amsterdam 1957, North Holland Publ. Co., Amsterdam, 1959, p. 158.
D. AXENTE, O. PIRINGER, J. Inorg. Nucl. Chem., 33 (1971) 665.
S. JORDAN, F. T. BONNER, Inorg. Chem., 12 (1973) 1369.
M. J. STERN, LOIS NASH KAUDER, W. SPINDEL, J. Chem. Phys., 36, 3 (1962) 764.
E. ABEL, H. SCHMIDT, S. BABAD, Z. Phys. Chem., 136 (1928) 135.
E. ABEL, H. SCHMIDT, Z. Phys. Chem., 134 (1928) 279; 136 (1928) 430.
D. AXENTE, O. PIRINGER, J. Inorg. Nucl. Chem., 34 (1972) 847.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Axente, D., Abrudean, M. & Bâldea, A. Nitrogen isotope exchange between nitric oxide and nitric acid. Journal of Radioanalytical and Nuclear Chemistry, Articles 207, 163–170 (1996). https://doi.org/10.1007/BF02036536
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02036536