Abstract
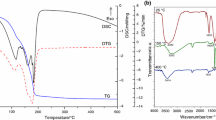
The thermochemical reduction of a series of structurally and morphologically different natural and synthetic manganese(IV) oxides has been investigated. Measurements have been performed by means of combined thermogravimetry/mass spectrometry, X-ray diffraction and analytical scanning electron microscopy. The mechanisms of the degradation of these materials have been characterized in order to establish standardized procedures for their reactivity as function of structure, morphology and experimental conditions. The corresponding results are the fundament with respect to a reproducible technical application.
Similar content being viewed by others
References
R. G. Burns and V. M. Burns, Rev. Min., 6 (1979) 1.
R. Giovanoli, Chem. Erde, 44 (1985) 227.
P. Ruetschi, J. Electrochem. Soc., 131 (1984) 2737.
Ullmann's Encycl. Ind. Chem., 5th Ed., A16 (1990) 124.
A. J. Fatadi, Synth., 65 (1976) 65.
R. Giovanoli, K. Bernhard and W. Feitknecht, Helv. Chim. Acta, 51 (1968) 355.
K. Wurr, Diploma Thesis, University of Hamburg, 1993.
K. Wurr and A. Reller, Progr. of GEFTA Meeting, Leipzig, 1994.
F. Fillaux et al., J. Electrochem. Soc., 140 (1993) 585.
Gmelin's Handbuch der anorg. Chem., ‘Manganese’, Vol. 56 C1 (1973) 295.
R. Scholder and U. Protzer, Z. Anorg. Allg. Chem., 369 (1969) 313.
Author information
Authors and Affiliations
Additional information
The authors would like to thank Dr. R. Lenck for compositional analyses of the investigated compounds. The financial support of the Chemetall GmbH, Frankfurt, Germany as well as the Fond der Chemischen Industrie is also acknowledged.
Rights and permissions
About this article
Cite this article
Wurr, K., Reller, A. Thermochemical reactivity of manganese(IV) oxides in reducing atmosphere. Journal of Thermal Analysis 47, 339–348 (1996). https://doi.org/10.1007/BF01983975
Issue Date:
DOI: https://doi.org/10.1007/BF01983975