Abstract
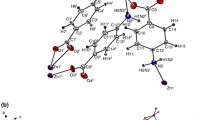
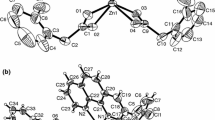
The new zinc(II) complexes of general formula Zn(CH3CH2CH2COO)2· nL (whereL = caffeine, nicotinamide, theobromine;n=1 or 2) were prepared and identified.
Thermal properties of these compounds were investigated by thermal analysis (TG/DTG, DTA, DSC/DDSC).
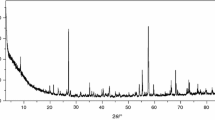
Gaseous products of thermal decomposition were detected by IR spectroscopy and Mass spectroscopy. Final products of thermal decomposition were determined by X-ray powder diffraction patterns.
Similar content being viewed by others
References
K. Györyová and V. Balek, J. Thermal Anal., 40 (1993) 519.
K. Györyová, V. Balek and J. Kovárová, Thermochim. Acta, (1995) in print.
E. Jóna, A. Sirota, M. Melník and M. Kubranová, Book of Abstracts TERMANAL'94 Vysoké Tatry, 1994, p. 133.
M. Melník, M. Anderová and M. Hol'ko, Inorg. Chim. Acta, 67 (1982) 117.
K. Györyová, M. Melník, J. Skorsepa and A. Eštoková, Contribution to development of Coord. Chem., 14th Conf. on Coord. Chem., Smolenice (Slovakia), 1993, p. 485.
Author information
Authors and Affiliations
Additional information
This work was supported by the Slovak Ministry of Education Grant No. 1/3230/96. This financial support is gratefully acknowledged.
Rights and permissions
About this article
Cite this article
Györyová, K., Balek, V., Behrens, B.H. et al. Thermal properties of zinc butyrate complex compounds. Journal of Thermal Analysis 48, 1263–1271 (1997). https://doi.org/10.1007/BF01983436
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01983436