Abstract
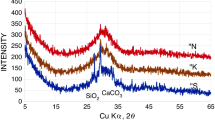
In the DTA studies of the clinkerization process the values of enthalpy attributed to the particular stages of clinker synthesis were determined and the energy consumption decrease due to the modification of the phase composition was calculated for 400 J/g.
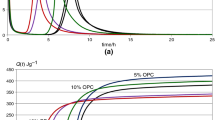
The activation of the low energy cement by Ba addition was shown using microcalorimetry and thermal methods.
Zusammenfassung
In einer DTA-Studie des Klinkerbildungsprozesses wurden die Enthalpiewerte der einzelnen Stufen der Klinkersynthese bestimmt. Die ermittelte Abnahme der Energieaufnahme für die Änderung der Phasenzusammensetzung beträgt 400 J/g.
Mittels Mikrokalorimetrie und thermischen Methoden konnte die Aktivierung des energetisch niedrig liegenden Zementes durch Zusatz von Ba nachgewiesen werden.
Access this article
We’re sorry, something doesn't seem to be working properly.
Please try refreshing the page. If that doesn't work, please contact support so we can address the problem.
Similar content being viewed by others
References
J. P. Meric, Ciment, Beton, Platre et Chaux, 1 (1987) 764.
J. Stark and A. Mueller, X IBAUSIL, Weimar 1988.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rajczyk, K., Nocun-Wczelik, W. Thermal methods and microcalorimetry application in the studies of low energy cements. Journal of Thermal Analysis 38, 771–775 (1992). https://doi.org/10.1007/BF01979407
Issue Date:
DOI: https://doi.org/10.1007/BF01979407