Abstract
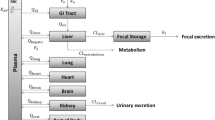
The single-dose toxicokinetics ofN-nitrosodimethylamine (NDMA) has been characterized in 8-week-old male Syrian golden hamsters by analysis using high performance liquid chromatography of serial blood samples. An i.v. bolus dose of 4.2 μmol/kg [14C]NDMA revealed biphasic first-order elimination with a terminal half-life of 8.7±1.0 min (mean±SE) for unchanged NDMA and 31.5±5.5 min for total radioactivity, and evidence for conversion to polar metabolites was seen in the chromatographic assays. The systemic blood clearance and apparent steady-state volume of distribution for unchanged NDMA were 51.2±3.0 ml/min/kg and 582±60 ml/kg, respectively. No unchanged NDMA was detected in the urine following an i.v. bolus dose of 15 μmol/kg [14C]NDMA, but 31% of the total radioactivity was eliminated by that route. A dose of 38 μmol/kg given by gavage indicated a systemic bioavailability of 11±4% for unchanged NDMA. Reversible binding of NDMA to hamster plasma proteins was found to be negligible. Estimation of the intrinsic hepatic clearance (ClI) in the hamster produced a value of 648 ml/min/kg, which is greater than that previously obtained for the rat, and indicates that the metabolic capacity of the hamster liver is greater than that of the rat. These results suggest that this difference in ClI may play a role in the previously reported (Lijinsky et al. 1987) switch in organotropism from almost exclusivity for liver tumors in hamsters dosed by gavage to additional high incidences of lung and kidney tumors in the rat.
Similar content being viewed by others
Abbreviations
- NDMA:
-
N-nitrosodimethylamine
- [14C]NDMA:
-
N-nitrosomethyl ([14C]methyl)amine
- i.g.:
-
intragastric
- A and B:
-
coefficients of the exponential terms which describe the change in blood concentration over time
- α and β:
-
first order rate constants for the distribution, elimination or absorption phases
- t 1/2(α) and t 1/2(β):
-
half-lives of the distribution, elimination, or absorption phases
- AUC:
-
area under the blood concentrationversus time curve
- Cl:
-
systemic blood clearance
- ClI :
-
intrinsic hepatic clearance
- F:
-
systemic bioavailability
- MRT:
-
mean residence time
- MAT:
-
mean absorption time
- Vss :
-
apparent steady-state volume of distribution
- HPLC:
-
high performance liquid chromatography
References
Adams JD, LaVoie EJ, Hoffmann D (1985) On the pharmacokinetics of tobacco-specific N-nitrosamines in Fischer rats. Carcinogenesis 6: 509–512
Altman PL, Dittmer DS (1974) Biology data book, 2nd edn, vol III. Federation of American Societies for Experimental Biology, Bethesda
Boxenbaum H (1980) Interspecies variation in liver weight, hepatic blood flow, and antipyrine intrinsic clearance: extrapolation of data to benzodiazepines and phenytoin. J Pharmacokinet Biopharm 8: 165–176
Denkel E, Pool BL, Schlehofer JR, Eisenbrand G (1986) Biological activity ofN-nitrosodiethanolamine and of potential metabolites which may arise after activation by alcohol dehydrogenase inSalmonella typhimurium, in mammalian cells, and in vivo. J Cancer Res Clin Oncol 111: 149–153
Diaz Gomez MI, Swann PF, Magee PN (1977) The absorption and metabolism in rats of small oral doses of dimethylnitrosamine in human food. Biochem J 164: 497–500
Garland WA, Kuenzig W, Ribio F, Kornychuk H, Norkus EP, Conney AH (1986) Urinary excretion of nitrosodimethylamine and nitrosoproline in humans: interindividual and intraindividual differences and the effect of administered ascorbic acid and α-tocopherol. Cancer Res 46: 5392–5400
Gombar CT, Pylypiw HM, Harrington GW (1987) Pharmacokinetics ofN-nitrosodimethylamine in beagles. Cancer Res 47: 343–347
Gombar CT, Harrington GW, Pylypiw HM, Bevill RF, Thurmon JC, Nelson DR, Magee PN (1988) Pharmacokinetics ofN-nitrosodimethylamine in swine. Carcinogenesis 9: 1351–1354
Gough TA, Webb KS, Swann PF (1983) An examination of human blood for the presence of volatile nitrosamines. Food Chem Toxicol 21: 151–156
Harrington GW, Magee PN, Pylypiw HM, Bevill RF, Nelson DR, Thurmon JC (1987) The pig as an animal model for the study of nitrosamine metabolism. In: Bartsch H, O'Neill IK, Schulte-Hermann R (eds) Relevance ofN-nitroso compounds to human cancer: exposures and mechanisms. IARC Scientific Publications, no 84, International Agency for Research on Cancer, Lyon, pp 132–134
Heath DF (1962) The decomposition and toxicity of dialkylnitrosamines in rats. Biochem J 85: 72–91
Keefer LK, Garland WA, Oldfield NF, Swagzdis JE, Mico BA (1985) Inhibition ofN-nitrosodimethylamine metabolism in rats by ether anesthesia. Cancer Res 45: 5457–5460
Kraft PL, Skipper PL, Charnley G, Tannenbaum SR (1981) Urinary excretion of dimethylnitrosamine: a quantitative relationship between dose and urinary excretion. Carcinogenesis 2: 609–612
Lijinsky W, Kovatch RM, Riggs CW (1987) Carcinogenesis by nitrosodialkylamines and azoxyalkanes given by gavage to rats and hamsters. Cancer Res 47: 3968–3972
Lutz WK (1984) Structural characteristics of compounds that can be activated to chemically reactive metabolites: use for a prediction of a carcinogenic potential. Arch Toxicol 7 [suppl]: 194–207
Magee PN (1956) Toxic liver injury: the metabolism of dimethylnitrosamine. Biochem J 64: 676–682
Magee PN (1985) Role of pharmacokinetics and DNA dosimetry in relatingin vitro andin vivo actions of N-nitroso compounds. Carcinog Compr Surv 10: 109–122
Mico BA, Swagzdis JE, Hu HS-W, Keefer LK, Oldfield NF, Garland WA (1985) Low-dosein vivo pharmacokinetics and deuterium isotope effect studies ofN-nitrosodimethylamine in rats. Cancer Res 45: 6280–6285
Pegg AE, Perry W (1981) Alkylation of nucleic acids and metabolism of small doses of dimethylnitrosamine in the rat. Cancer Res 41: 3128–3132
Preussmann R, Stewart BW (1984)N-Nitroso carcinogens. In: Searle CE (ed) Chemical carcinogens, 2nd edn, vol 2. American Chemical Society, Washington, pp 643–828
Raineri R, Poiley JA, Andrews AW, Pienta RJ, Lijinsky W (1981) Greater effectiveness of hepatocyte and liver S9 preparations from hamsters than rat preparations in activating N-nitroso compounds to metabolites mutagenic toSalmonella. J Natl Cancer Inst 67: 1117–1122
Streeter AJ, Nims RW, Hrabie JA, Heur Y-H, Keefer LK (1989) Sex differences in the single-dose toxicokinetics ofN-nitrosomethyl-(2-hydroxyethyl)amine in the rat. Cancer Res 49: 1783–1789
Streeter AJ, Nims RW, Sheffels PR, Heur Y-H, Yang CS, Mico BA, Gombar CT, Keefer LK (1990) Metabolic denitrosation ofN-nitrosodimethylaminein vivo in the rat. Cancer Res 50: 1144–1150
Swann PF (1982) Metabolism of nitrosamines: observations on the effect of alcohol on nitrosamine metabolism and on human cancer. Banbury Rep 12: 53–68
Swann PF, Coe AM, Mace R (1984) Ethanol and dimethylnitrosamine and diethylnitrosamine metabolism and disposition in the rat. Possible relevance to the influence of ethanol on human cancer incidence. Carcinogenesis 5: 1337–1343
Wishnok JS, Rogers AE, Sanchez O, Archer MC (1978) Dietary effects on the pharmacokinetics of three carcinogenic nitrosamines. Toxicol Appl Pharmacol 43: 391–398
Wishnok JS, Tannenbaum SR, Fox JG, Mesina JE, Hotaling LC (1987) The ferret as a model for endogenous synthesis and metabolism ofN-nitrosamines. In: Bartsch H, O'Neill IK, Schulte-Hermann R (eds) Relevance ofN-nitroso compounds to human cancer: exposures and mechanisms. IARC Scientific Publications, no 84, International Agency for Research on Cancer, Lyon, pp 135–137
Yoo J-SH, Ning SM, Patten CJ, Yang CS (1987) Metabolism and activation ofN-nitrosodimethylamine by hamster and rat microsomes: comparative study with weanling and adult animals. Cancer Res 47: 992–998
Yoo J-SH, Guengerich FP, Yang CS (1988) Metabolism ofN-nitrosodimethylamine by human liver microsomes. Cancer Res 48: 1499–1504
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Streeter, A.J., Nims, R.W., Wu, P.P. et al. Toxicokinetics ofN-nitrosodimethylamine in the Syrian golden hamster. Arch Toxicol 64, 562–566 (1990). https://doi.org/10.1007/BF01971835
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01971835