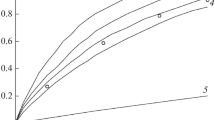
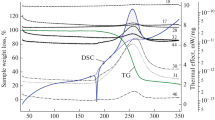
Abstract
By means of non-isothermal DTA in its simple form the initial exotherm temperaturesT D of 2,4-dinitrotoluene, 2,6-dinitrotoluene, 2,4,6-trinitrotoluene, 1,5-dinitronaphtalene, 1,8-dinitronaphthalene and 1,4,5,8-tetranitronaphthalene have been determined. By application of the Piloyan method to these measurements, the activation energiesE arising from the decompositions of these compounds were established. The results are discussed from the point of view of the molecular structures of the polynitro compounds.
Résumé
On a déterminé par ATD les températures initialesT D des phénomènes exothermiques du 2,4-dinitrotoluène, 2,6-dinitrotoluène, 2,4,6-trinitrotoluène, 1,5-dinitronaphtalène, 1,8-dinitronaphtalène, 1,4,5-trinitronaphtalène et 1,4,5,8-tétranitrophtalène. Les énergies d'activationE de la décomposition de ces composés ont été déterminées à l'aide de la mé thode de Piloyan. Les résultats obtenus sont discutés sous l'aspect de la structure moléculaire de ces composés.
Zusammenfassung
Durch nicht-isotherme DTA in ihrer einfachen Form wurde der BeginnT D der Exothermen von 2,4-Dinitrotoluol, 2,6-Dinitrotoluol, 2,4,6-Trinitrotoluol, 1,5-Dinitronaphthalin, 1,8-Dinitronaphthalin, 1,4,5-Trinitronaphthalin und 1,4,5,8-Tetranaphthalin bestimmt. Durch Anwendung der Methode von Piloyan auf diese Messungen wurden die aus der Zersetzung o.a. Verbindungen herrührenden AktivierungsenergienE spezifiziert. Die erhaltenen Ergebnisse werden unter dem Aspekt der Molekularstruktur der untersuchten Polynitroverbindungen erörtert.
Резюме
Посердством неизоте рмического ДТА в его простой форме были оп ределены начальныеT D эксотерм 2,4-динитротолуола, 2,6-динитротолуола, 2,4,6-три нитротолуола, 1,5-и 1,8-динитронафталино в, 1,4,5-тринитронафталин а и 1,4,5,8-тетранитронафтали на. Применяя к этим измер ениям метод Пилояна, б ыли установлены энергии активацииE реакций разложения э тих соединений. Получ енные результаты обсужден ы с точки зрения молекулярной структ уры изученных полинитросоединени й
Similar content being viewed by others
References
S. Zeman, J. Thermal Anal., 17 (1979) 19.
Yu. Ya. Maksimov, Zhur. Fiz. Khim., 46 (1972) 1726.
Yu. Ya. Maksimov, Zhur. Fiz. Khim., 45 (1971) 793.
Y. Hara andH. Osada, Kogyo Kayaku, 35 (1974) 26.
W. R. Deason, Ind. Eng. Chem., 51 (1959) 1001.
G. Krien, Explosivstoffe, 13 (1965) 205.
M. Itoh, T. Yoshida, M. Nakamura andK. Uetake, Kogyo Kayaku, 38 (1977) 17.
R. M. Gidry andL. P. Davis, Thermochim. Acta, 32 (1979) 1.
S. Zeman, Thermostable Polynitroaromatic Compounds. Part II. Res. Rep., PO 2-79, Úrad pro vynálezy a objevy, Prague, Jan. 1979.
S. Zeman, Thermostable Polynitroaromatic Compounds. Part I. Ph. D. Thesis, Univ. Chem. Technol., Pardubice, June 1973.
S. Zeman, J. Thermal Anal., 19 (1980) 207.
E. R.Ward, C. D.Johnson and L. A.Day, J. Chem. Soc., (1959) 487.
S. Zeffan andE. Zemanová, J. Thermal Anal., 19 (1980) 417.
S.Zeman and E.Zemanová, J. Thermal Anal., — this paper, Part VI.
S.Zeman and E.Zemanová, J. Thermal Anal., — this paper, Part VII.
V. G.Matveev and G. M.Nazin, Izv. Akad. Nauk SSSR, Ser. Khim. (1978) 771.
V. G.Matveev, V. V.Dubikhin and G. M.Nazin, Izv. Akad. Nauk SSSR, Ser. Khim. (1978) 474.
S. Zeman J. Thermal Anal., 19 (1980) 99.
A. G. Turovec andV. I. Danilova, Izv. Vysshikh Ucheb. Zaved., Fizika (1973) No. 08, 68.
M. J. Kamlet, J. C. Hoffsommer andH. G. Adolph, J. Am. Chem. Soc., 84 (1962) 3925.
H. G. Adolph, B. Johnson andM. J. Kamlet, J. Org. Chem., 30 (1965) 2864.
M. J. Kamlet, J. C. Hoffsommer, R. R. Minessinger andH. G. Adolph, J. Org. Chem., 33 (1968) 3070.
Bo Lamm andK. Nordfält, Acta Chem. Scand., 20 (1966) 1208.
V. F. Sapranovich, Yu. Ya. Maksimov andM. F. Makrelova, Trudy MCHTI im. Mendeleeva, 75 (1973) 147.
J. R.Holden and C.Dickinson, Chem. Commun. (1969) 114.
S. Zeman, Thermochim. Acta, 31 (1979) 269.
E. F. M.Britain, C. H. J.Wells, H. M.Paisley and D. J.Stickley, J. Chem. Soc. (B 1970) 1714.
V. G.Matveev, V. V.Dubikhin and G. M.Nazin, Izv. Akad. Nauk SSSR, Ser. Khim. (1978) 783.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zeman, S. Possibilities of applying the Piloyan method of determination of decomposition activation energies in the differential thermal analysis of polynitroaromatic compounds and their derivatives. Journal of Thermal Analysis 21, 9–14 (1981). https://doi.org/10.1007/BF01913694
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01913694