Abstract
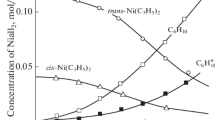
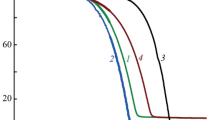
Thermal dissociation processes ofβ-diketonate adducts with the composition ML2 · 2B (L = acetylacetone and its substituted derivatives;M = Ni, Co;B = H2O, BuNH2CH3OH, Py, NH3) were investigated. Kinetics of dissociation were studied in a flow reactor. For dehydration processes a compensation relationship is observed (lgA=E+b), demonstrating analogous mechanisms.
A semi-quantitative series of the thermodynamic stabilities of the dihydrates is obtained from experimental data at quasi-equilibrium conditions (Q-derivatograph).
Zusammenfassung
Es wurden die thermischen Dissoziationsprozesse vonβ-Diketonataddukten der Zusammensetzung ML2 · 2B (L=Azetylazeton und dessen substituierte Abkömmlinge;M=Ni, Co;B=H2O, BuNH2, CH3OH, Py, NH3) untersucht. Die Kinetik der Dissoziation wurde in einem Flowreaktor untersucht. Als Beweis für die Ähnlichkeit der Mechanismen der Dehydrierungsprozesse konnte eine lgA=aE+b Beziehung festgestellt werden. Von unter Quasi-Gleichgewichtsbedingungen gewonnenen Daten konnte eine semiquantitative Serie der thermodynamischen Stabilitäten der Dihydrate abgeleitet werden (Q-Derivatograph).
Резюме
Изучены процессы тер мической диссоциаци и аддуктовβ-дикетонат ов состава ML2·2B (L = ацетилацетон и ег о замещенные произво дные;M= Ni, Co;B=H2O, BuNH2, CH3OH, Py, NH3). Кинетика диссоци ации исследована в проточном реакторе; д ля процессов дегидратации наблюд ается компенсационн ая зависимость (lgA=aEs+b) свиде тельствующая об аналогии в механиз ме. Полуколичественн ый ряд термодинамической у стойчивости дигидра тов получен по данным экс перимента в квазирав новесных условиях (Q-дериватогр аф).
Similar content being viewed by others
References
M. Z. Gurevich, T. M. Sas, N. E. Lebedeva, V. V. Zelentsov and B. D. Stepin, Zh. neorg. khim., 14 (1972) 1073.
M. Z. Turevich, T. M. Sas, V. B. Zelentsov, B. D. Stepin and N. E. Mazepova, Zh. neorg. khim., 20 (1975) 452.
E. A. Mazurenko, Zh. N. Bublik and S. V. Volkov, Ukr. khim. zs., 45 (1979) 591.
A. Aikawa, H. Kawamoto and M. Abe, J. Inorg. Nucl. Chem., 34 (1972) 1763.
J. Yoshida, H. Kobayashi and K. Ueno, J. Inorg. Nucl. Chem., 35 (1973) 4061.
J. Yoshida, H. Kobayashi and K. Ueno, Bull. Chem. soc. Japan, 45 (1972) 1411.
J. H. van Fassel and W. W. Wendlandt, J. Amer. Chem. Soc., 82 (1960) 4821.
W. W. Wendlandt, J. L. Berg and G. R. Norton, J. Phys. Chem., 64 (1960) 1289.
A. G. Jess, A. Baks, D. L. Stufkens and K. Vriese, Inorg. Chim. Acta, 29 (1978) 177.
M. A. Bernard, A. Busnot and N. Decker, Bull. Soc. Chim. France, 6 (1971) 1995.
M. A. Bernard, M. Borel and N. Decker, Bull. Soc. Chim. France, 9 (1971) 3144.
M. A. Bernard and N. Decker, Bull. Soc. Chim. France, 4 (1972) 1288.
M. A. Bernard and F. Busnot, Bull. Soc. Chim. France, 9 (1969) 3061.
M. A. Bernard, A. Busnot and F. Busnot, Bull. Soc. Chim. France, 8 (1970) 2867.
I. K. Igumenov, V. M. Gorbachev and A. N. Popov, Izv. Sibirsk. otd. AN SSSR, ser. khim. nauk, (1973) ser. 2. No. 4, 33.
J. Paulik and F. Paulik, Anal. Chim. Acta 60 (1972) 127.
F. Paulik and J. Paulik, J. Thermal Anal., 5 (1973) 253.
V. A. Logvinenko, Thermal analysis of co-ordination compounds and clathrates (in Russian). Nauk, Novosibirsk, 1982.
A. V. Nikolaev, V. A. Logvinenko, Ya. Šesták and V. Škvara, Dokl. AN SSSR, 231 (1976) 146.
H.H. Horowitz and G. Metzger, Anal. Chem., 35 (1963) 1464.
N. E. Fedotova and I. K. Igumenov, Izv. Sibirsk. Otdel. AN SSSR, ser. khim. nauk. (1984) ser. 3. No. 8, 20.
I. K. Igumenov, N. E. Fedotova and S. V. Zemskov, Koord. khimiya, 6 (1980) 1903.
I. K. Igumenov and A. N. Popov, Izv. Sibirsk. Otdel. AN SSSR, ser. khim. nauk, (1972) No. 5, 15.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Logvinenko, V.A., Fedotova, N.E., Igumenov, I.K. et al. Study of the thermal dissociation of processes of nickel(II) and cobalt(II) β-diketonate adducts. Journal of Thermal Analysis 34, 259–268 (1988). https://doi.org/10.1007/BF01913392
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01913392