Summary
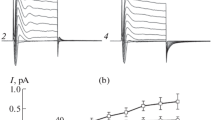
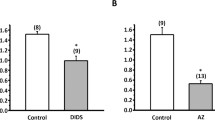
During their flux through the skin of the frogLeptodactylus ocellatus, Na+ and Cl− interact with each other. This interaction gives rise to electrical phenomena which are studied in the present paper. The skin is mounted in Na2SO4 Ringer's with 115 mM Na+ on the inside, and a variety of outer solutions,. The osmolarity of all solutions is kept constant at 237.8 mosmol by adding sucrose. When the main anion used on the outside is SO =4 the electrical potential difference (Δψ) rises steadily with the concentration of sodium (Na+)o up to 87 mV, which is reached at about 20mm. Thereafter Δψ remains constant. When the main anion is Cl− it is observed that Δψ rises steadily with (NaCl)o with a slope similar to the curve obtained with SO =4 (37 mV per decade), but with a lower intercept attributed to an inward Cl pumping which is characteristic of this frog species. At 2–9 mM (NaCl)o a Cl-specific channel is activated. Further increases of (NaCl)o produce a decrease of Δψ. The specificity of the activation of this site by monovalent cations and its use by monovalent anions is also studied.
Similar content being viewed by others
References
Cereijido, M., Curran, P.F. 1965. Intracellular electrical potentials in frog skin.J. Gen. Physiol. 48:543
Cereijido, M., Herrera, F.C., Flanigan, W., Curran, P.F. 1964. The influence of Na concentration on Na transport across frog skin.J. Gen. Physiol. 47:879
Di Bona, D.R., Civan, M.M. 1973. Pathways for movement of ions and water across toad urinary bladder. I. Anatomic site of transepithelial shunt pathways.J. Membrane Biol. 12:101
Dobson, J.G., Jr., Kidder, G.W., III 1968. Edge damage effect “in vitro” frog skin preparations.Am. J. Physiol. 214:719
Eisenman, G. 1962. Cation selective glass electrodes and their mode of operation.Biophys. J. 2:259
Finn, A.L. 1974. Transepithelial, potential difference in the urinary bladder is not due to ionic diffusion.Nature (London) 250:495
Fischbarg, J., Zadunaisky J.A., Fisch, F.W. 1967. Dependence of sodium and chloride transport on chloride concentration in isolated frog skin.Am. J. Physiol. 213:963
Franz, T.J., Van Bruggen, J.T. 1967. Hyperosmolarity and the net transport of nonelectrolytes in frog skin.J. Gen. Physiol. 50:933
Greven, K. 1944. Ein Beitrag zum Problem des Ruhestroms der Froschhaut.Pfluegers Arch. 244:365
Helman, S.I., Miller, D.A. 1973. Edge damage effect on electrical measurement of frog skin.Am. J. Physiol. 225:972
Huf, E.G. 1972. The role of Cl and other anions in active Na transport in isolated frog skin.Acta Physiol. Scand. 84:366
Kidder, G., Cereijido, M., Curran P.F. 1964. Transient changes in electrical potential differences across frog skin.Am. J. Physiol. 207:935
Koefoed-Johnsen, V., Ussing, H.H. 1958. The nature, of the frog skin potential.Acta Physiol. Scand. 42:298
Leb, D.E., Edwards, C., Lindley, B.D., Hoshiko, T. 1965. Interaction between the effects of inside and outside Na and K on Bullfrog skin potential.J. Gen. Physiol. 49:309
Linderholm, H. 1954. On the behaviour of the “sodium pump” in frog skin at various concentrations of Na ions in the solution on the epithelial side.Acta Physiol. Scand. 31:36
Lindley, B.D., Hoshiko, T. 1964. The effects of alkali metal-cations and common anions on the frog skin potential.J. Gen. Physiol. 47:749
Mandel, L.J., Curran, P.F. 1972. chloride flux via a shunt pathway in frog skin: Apparent exchange diffusion.Biochim. Biophys. Acta 282:258
Moreno, J.H., Reisin, I., Rodriguez Boulan, E., Rotunno, C.A., Cereijido, M. 1973. Barriers to Na movement across frog skin.J. Membrane Biol. 11:99
Ques-von Petery, M.V., Rotunno, C.A., Cereijido, M. 1978. Studies on chloride permeabilities of the skin ofLeptodactylus ocellatus: I. Na+ and Cl− effect on passive movements of Cl−.J. Membrane Biol. 42:317
Rabito, C.A., Rodriguez Boulan, E., Cereijido, M. 1973. Effect of the composition of the inner bathing solution on transport properties of the frog skin.Biochim. Biophys. Acta 311:630
Reuss, L., Finn, A. 1974. Passive electrical properties of toad urinary bladder epithelium. Intercellular electrical coupling and transepithelial cellular shunt conductance.J. Gen. Physiol. 64:1
Rotunno, C.A., Ques-von Petery, M.V., Cereijido, M. 1978. Studies on chloride permeabilities of the skin ofLeptodactylus ocellatus: II. Na+ and Cl− effect on inward movements of Cl−.J. Membrane Biol. 42:331
Smith, T.C., Hughes, W.D., Huf, E.G. 1971. Movement of CO2 and CO3H− across isolated frog skin.Biochim. Biophys. Acta 225:77
Smith, T.C., Martin, J.H., Huf, E.F. 1973. Na pool and Na concentration in epidermis of frog skin.Biochim. Biophys. Acta 291:465
Steinbach, H.B. 1933. The electrical potential difference across living frog skin.J. Cell Comp. Physiol. 3:1
Urakabe, S., Handler, J.S., Orloff, J. 1970. Effect of hypertonicity on permeability properties of the toad bladder.Am. J. Physiol. 218:1179
Ussing, H.H., Windhager, E.E. 1964. Nature of shunt path and active sodium transport path through frog skin epithelium.Acta Physiol. Scand. 61:484
Wade, J.B., Revel, J.P., Di Scala V.A. 1973. Effect of osmotic gradients on intercellular junctions of the toad bladder.Am. J. Physiol. 224:407
Zadunaisky, J.A., Candia, O.A. 1962. Active transport of sodium and chloride by the isolated skin of the South American frogLeptodactylus ocellatus.Nature (London) 195:1004
Zadunaisky, J.A., Candia, O.A., Chiarandini, D.J. 1963. The origin of the short-circuit current in the isolated skin of the South American frogLeptodactylus ocellatus.J. Gen. Physiol. 47:393
Zadunaisky, J.A., Fisch, F.W. 1964. Active and passive chloride movements across isolated amphibian skin.Am. J. Physiol. 207:1010
Zylber, E.A., Rotunno, C.A., Cereijido, M. 1973. Ion and water balance in isolated epithelial cells of the abdominal skin of the frogleptodactylus ocellatus.J. Membrane Biol. 13:199
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rodríguez. Boulan, E., Ques-von Petery, M.V., Rotunno, C.A. et al. Studies on chloride permeability of the skin ofLeptodactylus ocellatus: III. Na+ and Cl− effect on electrical phenomena. J. Membrain Biol. 42, 345–356 (1978). https://doi.org/10.1007/BF01870355
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01870355