Abstract
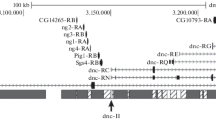
A minichromosome has originated from the transposing elementTE1. This autonomously replicating chromosome contains the structural genes white and roughest, from theDrosophila X chromosome. It arose within a stock carryingTE1 at 45F on chromosome2. In addition to thew andrst genes, the minichromosome may carry section 45C–45F from chromosome2. It is inherited by 33%–47% of the offspring. By this criterion it carries a centromere, although the origin of the centromere is unknown. From this minichromosome a still smaller one has originated, probably through the loss of all material from chromosome2 together with some heterochromatin. At the same time a duplication of white and roughest could have taken place. This chromosome has a strange morphology and is more frequently lost in meiosis than the larger one, but is still transmitted to about 29%–37% of the progeny of one parent heterozygous for the minichromosome. In both cases the flies have variegated eyes, probably as a result of position-effect variegation. The variegation pattern is influenced by factors in theX chromosome. The size of the smaller minichromosome is little more than 1 Mb as determined by pulsed field gel electrophoresis.
Similar content being viewed by others
References
Awad AAM, Bencze G, Gausz J (1984) The effect of X-rays and EMS on the behavior of the transposing element, TE98, inDrosophila melanogaster. Experientia 90:744–745
Collins M, Rubin GM (1984) Structure of chromosomal rearrangements induced by the FB transposable element inDrosophila. Nature 308:323–327
Dimitri P, Pisano C (1989) Position effect variegation inDrosophila melanogaster: relationship between suppression effect and the amount of Y chromosome. Genetics 122:793–800
Goldberg ML, Paro R, Gehring WJ (1982) Molecular cloning of the white locus region ofDrosophila melanogaster using a large transposable element. EMBO J 1:93–98
Green MM, Yamamoto M-T, Miklos GLB (1987) Genetic instability inDrosophila melanogaster: Cytogenetic analysis of MR-induced X-chromosome deficiencies. Proc Natl Acad Sci USA 84:4533–4537
Grell RF (1964a) Distributive pairing: The size-dependent mechanism for regular segregation of the fourth chromosomes inDrosophila melanogaster. Proc Natl Acad Sci USA 52:226–232
Grell RF (1964b) Chromosome size at distributive pairing inDrosophila melanogaster females. Genetics 50:151–166
Gubb D, Roote J, McGill S, Shelton M, Ashburner M (1986) Interactions between white genes carried by a large transposing element and the zeste1 allele inDrosophila melanogaster. Genetics 112:551–575
Hägele K, Ranganath HA (1987) The microchromosomes of five members of theDrosophila nasuta subgroup. Dros Inf Serv 66:72–73
Ising G, Block K (1981) Derivation-dependent distribution of insertion sites for aDrosophila transposon. Cold Spring Harbor Symp Quant Biol 45:527–544
Ising G, Block K (1984) A transposon as a cytogenetic marker inDrosophila melanogaster. Mol Gen Genet 196:6–16
Ising G, Ramel C (1976) The behaviour of a transposing element inDrosophila melanogaster. In: Ashburner M, Novitski E (eds) The genetics and biology ofDrosophila, vol. 1b. Academic Press, New York, pp 927–954
Kodani M, Stern C (1946) An “invisible” chromosome. Science 104:620–621
Langer-Safer PR, Levine M, Ward DC (1982) Immunological method for mapping genes onDrosophila polytene chromosomes. Proc Natl Acad Sci USA 79:4381–4385
Lefevre G (1976) A photographic representation and interpretation of the polytene chromosomes ofDrosophila melanogaster salivary glands. In: Ashburner M, Novitski E (eds) The genetics and biology of Drosophila, vol 1a. Academic Press, New York, pp 31–66
Lindsley D, Zimm G (1987) The genome ofDrosophila melanogaster. Part 3: rearrangements. Dros Inf Serv 65:71–74
Maniatis T, Hardison RC, Lacy E, Laner J, O'Connel C, Quon D, Sim GK, Efstradiatis A (1978) The isolation of structural genes from libraries of eukaryotic DNA. Cell 15:687–701
Paro R, Goldberg M, Gehring WJ (1983) Molecular analysis of a large transposable element carrying the white locus inDrosophila melanogaster. EMBO J 2:853–860
Reuter G, Gausz J, Gyurkovics H, Friede B, Bang R, Spierer A, Hall LMC, Spierer P (1987) Modifiers of position-effect variegation in the region from 86C to 88B of theDrosophila melanogaster third chromosome. Mol Gen Genet 210:429–436
Rubin G (1978) Isolation of a telomeric DNA sequence fromDrosophila melanogaster with a differential telomeric distribution. Cold Spring Harbor Symp Quant Biol 42:1041–1046
Sinclair D, Lloyd V, Grigliatti T (1989) Characterization of mutations that enhance position-effect variegation inDrosophila melanogaster. Mol Gen Genet 216:328–333
Snow R (1963) Alcoholic hydrochloric acid-carmine as a stain for chromosomes in squash preparations. Stain Technol 38:9–12
Southern EM (1975) Detection of specific sequences among DNA fragments separated by gel electrophoresis. J Mol Biol 98:503–517
Spofford JB (1976) Position effect variegation inDrosophila. In: Ashburner M, Nowitski E (eds) The genetics and biology ofDrosophila, vol 1c. Academic Press, New York, pp 955–1018
van der Bliek AM, van der Welde-Koerts T, Ling V, Borst P (1986) Overexpression and amplification of five genes in a multidrugresistant Chinese hamster ovary cell line. Mol Cell Biol 6:1671–1678
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Block, K., Ising, G. & Ståhl, F. Minichromosomes inDrosophila melanogaster derived from the transposing elementTE1 . Chromosoma 99, 336–343 (1990). https://doi.org/10.1007/BF01731721
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01731721