Summary
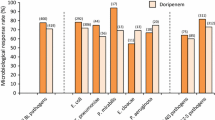
Fifty-five patients with gram-positive bacteremias were treated with cefotaxime after enrollment in comparative and non-comparative study protocols. Forty-nine of these 55 patients were evaluable and followed for their response to therapy and adverse effects. Most patients were white males 50 years of age or older (69%); 45% had two or more serious underlying diseases. Pneumonias caused 59% of these bacteremias, which were etiologically due toStreptococcus pneumoniae (22 episodes),Staphylococcus aureus (15), coagulase-negative staphylococci (3) and other streptococci (12). Overall, 90% of bacteremias were cured with cefotaxime therapy. Among five treatment failures were included three deaths, one due to cefotaxime-associated pseudomembranous colitis, one caused by a bacteremic superinfection due toPseudomonas aeruginosa and one due to a progressive pneumonia despite therapy. Adverse effects of therapy were infrequent and noteworthy for only one patient with questionable nephrotoxicity and a lack of cefotaxime-associated coagulopathy.
Zusammenfassung
55 Patienten mit grampositiven Bakteriämien wurden nach Zuordnung zu Studienprotokollen, die entweder für Vergleichsstudien konzipiert waren oder für nicht vergleichende Studien, mit Cefotaxim behandelt. Von diesen 55 Patienten standen 49 für eine Auswertung bezüglich Therapie-Erfolg und Nebenwirkungen zur Verfügung. Die meisten Patienten waren Kaukasier im Alter von 50 Jahren oder darüber (69%). 45% hatten zwei oder mehr ernsthafte Grundkrankheiten. 59% der Bakteriämien gingen von Pneumonien aus, die durchStreptococcus pneumoniae (22 Fälle),Staphylococcus aureus (15), koagulasenegative Staphylokokken (3) und andere Streptokokken (12) verursacht waren. Insgesamt wurden 90% der Bakteriämien unter Cefotaximtherapie geheilt. Unter den fünf Therapieversagern waren drei Todesfälle, von denen einer auf eine mit der Cefotaximbehandlung assoziierte pseudomembranöse Colitis zurückzuführen war, einer durchPseudomonas aeruginosa und einer durch die trotz Therapie fortschreitende Pneumonie bedingt war. Nebenwirkungen traten nur selten auf und sind nur bei einem Patienten mit fraglicher Nephrotoxizität erwähnenswert; eine Koagulopathie trat im Zusammenhang mit Cefotaximtherapie nicht auf.
Similar content being viewed by others
Literature
Counts, G. W., Turck, M. Antibacterial activity of a new parenteral cephalosporin — HR 756: comparison with cefamandole and ceforanide. Antimicrob. Agents Chemother. 16 (1979) 64–68.
Hamilton-Miller, J. M. T., Brumfitt, W., Reynolds, A. V. Cefotaxime (HR 756), a new cephalosporin with exceptional broad-spectrum activityin vitro. J. Antimicrob. Chemother. 4 (1978) 437–444.
Murray, P. R. Activity of cefotaxime-aminoglycoside combinations against aminoglycoside-resistantPseudomonas. Antimicrob. Agents Chemother. 17 (1980) 474–476.
Neu, H. C., Aswapokee, N., Aswapokee, P., Fu, K. P. HR 756, a new cephalosporin active against gram-positive and gram-negative aerobic and anaerobic bacteria. Antimicrob. Agents Chemother. 15 (1979) 273–281.
Sosna, J. P., Murray P. R., Medoff, G. Comparison of thein vitro activities of HR 756 with cephalothin, cefoxitin, and cefamandole. Antimicrob. Agents Chemother. 14 (1978) 876–879.
Mouton, R. P., Bongaerts, G. P. A., van Gestel, M. Comparison of activity and beta-lactamase stability of cefotaxime with those of six other cephalosporins. Antimicrob. Agents Chemother. 16 (1979) 757–760.
Peters, G., Pulverer, G. Comparativein vitro activity of cefotaxime (HR 756). Chemotherapy 26 (1980) 177–183.
Ramirez-Ronda, C. H., Nevarez, M., Saavedra, S., Bermudez, R. H. Comparativein vitro activity of moxalactam and cefotaxime againist isolates from blood culture. J. Antimicrob. Chemother. 7 (1981) 629–635.
Muytjens H. L., van der Ros-van de Repe, J. Comparative activities of 13 ß-lactam antibiotics. Antimicrob. Agents Chemother. 21 (1982) 925–934.
Neu, H. C. The new beta-lactamase-stable cephalosporins. Ann. Intern. Med. 97 (1982) 408–419.
Landesman, S. H., Corrado, M. L., Cherubin, C. E., Sierra, M. F. Activity of moxalactam and cefotaxime alone and in combination with ampicillin or penicillin aganinst group B streptococci. Antimicrob. Agents Chemother. 19 (1981) 794–797.
Cherubin, C. E., Corrado, M. L., Sierra, M. F., Gombert, M. E., Shulman, M. Susceptibility of gram-positive cocci to various antibiotics, including cefotaxime, moxalactam, andN-formimidoyl thienamycin. Antimicrob. Agents Chemother. 20 (1981) 553–555.
Moellering, R. C., Jr. Enterococcal infections in patients treated with moxalactam. Rev. Infect. Dis. 4 Suppl. (1982) S708-S711.
Yu, V. Enterococcal superinfection and colonization after therapy with moxalactam, a new broad spectrum antibiotic. Ann. Intern. Med. 94 (1981) 784–785.
Thornsberry, C., Sahm, D. F. Effect of media and blood on the antimicrobial activity of cephalosporins on serogroup D streptococci: a review. Diag. Microbial. Infect. Dis. 2 (1984) 75S-84S.
Francke, E. L., Neu, H. C. Use of cefotaxime, aβ-lactamase stable cephalosporin, in the therapy of serious infections, including those due to multiresistant organisms. Am. J. Med. 71 (1981) 435–442.
Schleupner, C. J., Engle, J. C. Clinical evaluation of cefotaxime for therapy of lower respiratory tract infections. Antimicrob. Agents Chemother. 21 (1982) 327–333.
Mullaney, D. T., John, J. F. Cefotaxime therapy. Evaluation of its effect on bacterial meningitis, CSF drug levels, and bactericidal activity. Arch. Intern. Med. 143 (1983) 1705–1708.
Meyers, B. R. Clinical experience with cefotaxime in the treatment of patients with bacteremia. Rev. Infect. Dis. 4 Suppl. (1982) S411-S415.
Madsen, P. O. Treatment of urinary tract infections with cefotaxime: noncomparative and prospective comparative trials. Rev. Infect. Dis. 4 Suppl. (1982) S416-S420.
Perkins, R. L. Clinical trials of cefotaxime for the treatment of bacterial infections of the lower respiratory tract. Rev. Infect. Dis. 4 Suppl. (1982) S421-S431.
Kemmerich, B., Lode, H., Belmega, G., Jendroschek, T., Borner, K., Koeppe, P. Comparative pharmacokinetics of cefoperazone, cefotaxime, and moxalactam. Antimicrob. Agents Chemother. 23 (1983) 429–434.
Schrinner, E., Limbert, M., Seeger, K., Seibert, G., Novick, W. J., Jr. Thein vitro antimicrobial activity of desacetyl-cefotaxime compared to other relatedβ-lactams. Diag. Microbiol. Infect. Dis. 2 (1984) 13S-20S.
Jones, R. N., Barry, A. L., Packer, R. R. The activity of cefotaxime and desacetylcefotaxime alone and in combination against anaerobes and staphylococci. Diag. Microbiol. Infect. Dis. 2 (1984) 37S-46S.
Bauer, A. W., Kirby, W. M. M., Sherris, J. C., Turck, M. Antibiotic susceptibility testing by a standardized single disk method. Am. J. Clin. Pathol. 45 (1966) 493–496.
Fuchs, P. C., Barry, A. L., Thornsberry, C., Jones, R. N., Gavan, T. L., Gerlach, E. H., Sommers, H. M. Cefotaxime:in vitro activity and tentative interpretive standards for disk susceptibility testing. Antimicrob. Agents Chemother. 18 (1980) 88–93.
Ericsson, H. M., Sherris, J. C. Antibiotic sensitivity testing. Report of an international collaborative study. Acta Pathol. Microbiol. Scand. Sect. B Suppl. (1971) 2–90.
Smith, C. R. Cefotaxime and cephalosporins: adverse reactions in perspective. Rev. Infect. Dis. 4 Suppl. (1982) S481-S488.
Thornsberry, C., Jones, R. N., Barry A. L., Fuchs, P. C. Antimicrobial susceptibility tests with cefotaxime and correlation with clinical bacteriologic response. Rev. Infect. Dis. 4 Suppl. (1982) S316-S324.
Ings, R. M. J., Fillastre, J. P., Godin, M., Leroy, A., Humbert, G. The pharmacokinetics of cefotaxime and its metabolites in subjects with normal and impaired renal function. Rev. Infect. Dis. 4 Suppl. (1982) S379-S391.
Wise, R., Wills, P. J., Andrews, J. M., Bedford, K. A. Activity of the cefotaxime (HR 756) desacetyl metabolite compared with those of cefotaxime and other cephalosporins. Antimicrob. Agents Chemother. 17 (1980) 84–86.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Schleupner, C.J. Clinical experience with cefotaxime for the therapy of bacteremias due to gram-positive organisms. Infection 13 (Suppl 1), S28–S33 (1985). https://doi.org/10.1007/BF01644214
Issue Date:
DOI: https://doi.org/10.1007/BF01644214