Summary
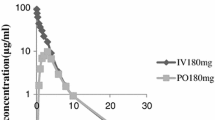
Pivampicillin hydrochloride (the only form at present widely available) is not suitable for administration to infants. Pivampicillin free base, however, can be used in syrup form and is therefore suitable for paediatric use. We have carried out a preliminary pharmacokinetic study in adult volunteers with the pivampicillin free base. The mean peak blood level following a dose equivalent to 250 mg. anhydrous ampicillin, attained after one hour, was 5.2 µg/ml.; mean urinary recovery was 72.2%. Thus, the free base is as well absorbed as is the hydrochloride.
Zusammenfassung
Pivampicillin-Hydrochlorid, die einzige zur Zeit verfügbare Form des Medikamentes, ist für die Anwendung bei Kindern ungeeignet. Die freie Base von Pivampicillin kann in Form eines Sirups angewendet werden und ist deshalb für den kinderärztlichen Gebrauch geeignet. Wir haben eine vorläufige pharmakokinetische Untersuchung mit der freien Pivampicillin-Base bei freiwilligen Erwachsenen durchgeführt. Die mittlere Spitzenkonzentration einer Dosis entsprechend 250 mg von wasserfreiem Ampicillin erreichte nach einer Stunde 5,2 µg/ml. 72,2% der 250 mg konnten im Urin wieder in aktiver Form nachgewiesen werden. Daraus ergibt sich, daß die freie Base so gut wie das Hydrochlorid resorbiert wird.
Similar content being viewed by others
References
von Daehne, W., Frederiksen, E., Gundersen, E., Lund, F., Morch, P., Petersen, H. J., Roholt, K., Tybring, L., Godtfredsen, W. O. Acyloxymethyl esters of ampicillin. J. med. Chem. 13 (1970) 607–612.
Marget, W., Dashner, F., Unertl, K. Investigations on pivampicillin treatment in newborns and infants. Infection 1 (1973) 41–45.
Marget, W., Wagner, M. Orale Anwendung von Propicillin, Ampicillin und Dicloxacillin im Säuglingsalter. Med. Klin. 62 (1967) 300–305.
Fernandez, C. A., Menezes, J. P., Ximenes, J. The effect of food on the absorption of pivampicillin and a comparison with the absorption of ampicillin potassium. J. int. Med. Res. 1 (1973) 530–533.
Foltz, E. L., West, J. W., Breslow, I. H., Wallick, H. Clinical pharmacology of pivampicillin. Antimicrob. Ag. Chemother. 1970 (1971) 442–454.
Brumfitt, W., Franklin, I., Hayek, L., Pursell, R. Treatment of urinary tract infection with pivampicillin, a new ampicillin derivative. Scand. J. inf. Dis. 5 (1973) 59–65.
Jordan, M. C., de Maine, J. B., Kirby, W. M. M. Clinical pharmacology of pivampicillin as compared with ampicillin. Antimicrob. Ag. Chemother. 1970 (1971) 438–441.
Hultberg, E. R., Backelin, B. Studies on the absorption of pivampicillin and ampicillin. Scand. J. inf. Dis. 4 (1972) 149–153.
von Daehne, W., Godtfredsen, W. O., Roholt, K., Tybring, L. Pivampicillin, a new orally active ampicillin ester. Antimicrob. Ag. Chemother. 1970 (1971) 431–437.
Knudsen, E. T., Rolinson, G. N., Stevens, S. Absorption and excretion of „Penbritin“. Brit med. J. 2 (1961) 198–200.
Poole, J. W., Owen, G., Silverio, J., Freyhof, J. N., Rosenman, S. B. Physico-chemical factors influencing the absorption of the anhydrous and trihydrate forms of ampicillin. Curr. therap. Res. 10 (1968) 292–303.
Wilcox, J. B., Brogden, R. N., Avery, G. S. Pivampicillin: a preliminary report of its pharmacokinetic properties and therapeutic efficacy. Drugs 6 (1973) 94–103.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hamilton-Miller, J., Kosmidis, J. & Brumfitt, W. Pharmacokinetic studies with a new pivampicillin: The free base. Infection 2, 193–195 (1974). https://doi.org/10.1007/BF01641460
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01641460