Abstract
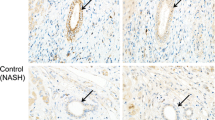
The proliferative activity and ultrastructural characteristics of proliferating biliary epithelial cells were analysed immunohistocytochemically in 39 biopsied liver specimens from patients with acute viral hepatitis, chronic hepatitis and liver cirrhosis using a monoclonal antibody against DNA polymerase α (DNA-PA). In acute viral hepatitis with perivenular confluent necrosis, proliferation of typical bile ducts was found frequently in portal areas. In chronic aggressive hepatitis and cirrhosis, ductular proliferation of both typical and atypical forms was found in enlarged portal and periportal areas and in confluent necrotic areas. The number of proliferating biliary epithelial cells that stained positive for DNA-PA was small. There were very few positively stained cells in atypical bile ducts in confluent necrotic areas of cirrhosis. Atypical bile ducts seen in chronic aggressive hepatitis, cirrhosis and acute hepatitis with confluent necrosis were positively stained for both cytokeratins 8 and 19. In cirrhosis, the number of stained biliary epithelial cells in typical bile ducts was larger than the number of such cells in atypical bile ducts (P< 0.01). By electron microscopy, the cells positively stained for DNA-PA were mostly so-called clear cells with irregular nuclei containing coarse nucleoplasm, and a few small cells with scanty cytoplasm and few organelles.
Similar content being viewed by others
References
Alpini G, Lenzi R, Zhai WR, Liu MH, Slott PA, Paronetto F, Tavoloni N (1989) Liver cell fraction enriched in cells with biliary epithelial phenotypes. Gastroenterology 97:1248–1260
Bensch KG, Tanaka S, Hu SZ, Wang TSH, Korn D (1982) Intracellular localization of human DNA polymeraseα with monoclonal antibodies. J Biol Chem 257:8391–8396
Bianchi L, Zimmer-Ning M, Gudat F (1979) Viral hepatitis. In: MacSween RMN, Anthony P, Scheuer P (eds) Pathology of the liver. Churchill Livingstone, New York, pp 164–191
Braun N, Papadopoulas T, Muller-Hermelink HK (1988) Cell cycle dependent distribution of the proliferation-associated Ki-67 antigen in human embryonic lung cells. Virchows Arch [B] 56:25–33
Buyssens N (1965) Ductular proliferation. Gastroenterology 49:702–706
Desmet VJ (1987) Modulation of biliary epithelium. In: Popper H, Arias IM, Heinrich PC, Keppler D, Landmann L (eds) Modulation of liver cell expression. MTP Press, Lancaster, pp 195–214
Eyken PV, Sciot R, Damme BV, Wolf-Peeters CD, Desmet VJ (1987) Keratin immunohistochemistry in normal human liver. Cytokeratin pattern of hepatocytes, bile ducts and acinar gradient. Virchows Arch [A] 412:63–72
Eyken PV, Sciot R, Desmet VJ (1989) A cytokeratin immunohistochemical study of cholestatic liver diseases: evidence that hepatocytes can express 'bile duct-type' cytokeratins. Histopathology 15:125–135
Garcia RL, Coltrera MD, Gown AM (1989) Analysis of proliferative grade using anti-PCNA cyclin monoclonal antibodies in fixed embedded tissues. Am J Pathol 134:733–739
Gerdes J, Lemke H, Baisch H, Wacker HH, Schwab U, Stein H (1984) Cell cycle analysis of a cell proliferation-associated human nuclear antigen defined by the monoclonal antibody Ki-67. J Immunol 133:1710–1715
Gratzner HG (1982) Monoclonal antibody to 5-bromo- and 5′-iododeoxyuridine: a new reagent for detection of DNA replication. Science 218:474–476
Grishman JW, Porta EA (1964) Origin and fate of proliferated hepatic ductal cells in the rat: electron microscopic and autoradiographic studies. Exp Mol Pathol 3:242–261
Kawakita N, Seki S, Sakaguchi H, Yanai A, Kuroki T, Mizoguchi Y, Kobayashi K, Monna T (1992) Analysis of proliferating hepatocytes using a monoclonal antibody against proliferating cell nuclear antigen (PCNA)/(cyclin) in embedded tissues from various liver diseases fixed in formaldehyde. Am J Pathol 140:513–520
Masaki S, Shiku H, Kaneda T, Koiwai D, Yoshida S (1982) Production and characterization of monoclonal antibody against DNA polymeraseα from calf thymus. Nucleic Acids Res 10:4703–4713
Masuko K, Rubin E, Popper H (1963) Proliferation of bile ducts in cirrhosis. Arch Pathol 78:421–431
Mushika M, Miwa T, Suzuoki Y, Hayashi K, Masaki S, Kaneda T (1988) Detection of proliferative cells in dysplasia, carcinoma in situ, and invasive carcinoma of uterine cervix by monoclonal antibody against DNA polymeraseα. Cancer 61:1182–1186
Nakamura T, Yoshimoto K, Nakayama Y, Tomita Y, Ichihara A (1983) Reciprocal modulation of growth and differentiated functions of mature rat hepatocytes in primary culture by cell-cell contact and cell membrane. Proc Natl Acad Sci USA 80:7229–7233
Nakamura H, Morita T, Masaki S, Yoshida S (1984) Intracellular localization and metabolism of DNA polymeraseα in human cells visualized with monoclonal antibody. Exp Cell Res 151:123–133
Namikawa R, Ueda R, Suchi T, Itoh G, Oto K, Takahashi T (1987) Double immunoenzymatic detection of surface phenotype of proliferating lymphocytes in situ with monoclonal anti-bodies against DNA polymeraseα and lymphocyte membrane antigens. Am J Clin Pathol 87:725–731
Phillips MJ, Poucell S, Patterson J, Valencia P (1987) The normal liver. The liver: an atlas and text of ultrastructural pathology. Raven Press, New York, pp 1–36
Popper H (1986) General pathology of the liver: light microscopic aspects serving diagnosis and interpretation. Semin Liver Dis 6:175–184
Sasaki H, Schaffner F, Popper H (1967) Bile ductules in cholestasis: morphologic evidence for secretion and absorption in man. Lab Invest 16:84–95
Schaffner F, Popper H (1961) Electron microscopic studies of normal and proliferated bile ducts. Am J Pathol 38:394–410
Seki S, Sakaguchi H, Kawakita N, Yanai A, Kim K, Mizoguchi Y, Kobayashi K (1990) Identification and fine structure of proliferating hepatocytes in malignant and nonmalignant liver diseases by a monoclonal antibody against DNA polymerase alpha. Hum Pathol 21:1020–1030
Seki S, Sakaguchi H, Kawakita N, Yanai A, Kuroki T, Mizoguchi Y, Kobayashi K, Monna T (1991) Detection of proliferating liver cells in various diseases by a monoclonal antibody against DNA polymerase a: with special reference to the relationship between hepatocytes and sinusoidal cells. Hepatology 14:781–788
Shimizu A, Tarao K, Takemiya S, Harada M, Inoue T, Ono T (1988) S-phase cells in diseased human liver determined by an in vitro BrdU-anti-BrdU method. Hepatology 8:1535–1539
Slott P, Liu M, Tavoloni N (1990) Origin, pattern, and mechanism of bile duct proliferation following biliary obstruction in the rat. Gastroenterology 99:466–477
Steiner JW, Carruthers JS (1963) Electron microscopy of hyperplastic ductular cells inα-naphthyl isothiocyanate-induced cirrhosis. Lab Invest 12:471–498
Stosiek P, Karsten U, Kasper K (1985) Monoclonal anti cytokeratin antibodies reveal bile duct origin of cirrhotic ductular proliferation (dp). Virchows Arch [A] 407:233–235
Takino T, Okanoue T, Kagawa K, Sugihara T, Sugino S, Ogasawara T, Masuda M (1976) Electron microscopic study on the proliferated biliary epithelial cells in fulminant hepatitis. J Clin Electron Microsc 9:5–6
Tanaka S, Hu SZ, Wang TSF, Korn D (1982) Preparation and preliminary characterization of monoclonal antibody against DNA polymeraseα. J Biol Chem 257:8391–8396
Uchida T, Peters RL (1983) The nature and origin of proliferated bile ductules in alcoholic liver disease. Am J Clin Pathol 79:326–333
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Seki, S., Sakaguchi, H., Kawakita, N. et al. Analysis of proliferating biliary epithelial cells in human liver disease using a monoclonal antibody against DNA polymeraseα . Vichows Archiv A Pathol Anat 422, 133–143 (1993). https://doi.org/10.1007/BF01607165
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01607165