Summary
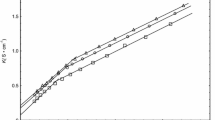
The effect of sodium chloride on the micellar properties of an anionic-nonionic detergent C16H33(OCH2·CH2)7OSO3Na has been investigated and the results have been compared with previous measurements on the nonionic analogue C16H33(OCH2·CH2)7OH. Light scattering and viscosity measurements showed that, over the electrolyte concentration range studied (0–1.0M NaCl), the micelles were very much smaller (m.m.w.=74,800 in 0.1M NaCl) than those of the nonionie analogue and exhibited no similar variation of size, shape or hydration with temperature, indicating that addition of electrolyte caused insufficient shielding of the micellar charge to induce nonionic behaviour. Viscosity results suggested a reduction in the micellar hydration with increase in electrolyte concentration to a limiting value of 0.28 g H2O/g of detergent for salt concentrations greater than 0.01M. A possible explanation of this effect is proposed. The effect of electrolyte on the c.m.c. was determined from surface tension measurements and was described by the equation, log c.m.c.=−6.4−0.54 log (c.m.c.+salt conc.) Comparison with data reported for sodium hexadecylpolyoxyethylene sulphates with shorter ethylene oxide chain lengths indicated a decrease in the c.m.c. in salt-free systems with increase in the chain length, the data obeying the equation, log c.m.c.=−3.5−0.20n wheren is the number of ethylene oxide groups in the chain.
Similar content being viewed by others
References
Bistline, R. G., A. J. Stirton, J. K. Weil, andE. W. Maurer, J. Amer. Oil Chemists' Soc.34, 516 (1957).
Weil, J. K., R. G. Bistline, andA. J. Stirton, J. Phys. Chem.62, 1083 (1958).
Weil, J. K., A. J. Stirton, R. G. Bistline, andE. W. Maurer, J. Amer. Oil Chemists' Soc.36, 241 (1959).
von Gotte, E., 3rd International Congress of Surface Activity, Vol.1, p. 45 (1960).
Schick, M. J., J. Phys. Chem.67, 1796 (1963).
Tokiwa, F. andK. Ohki, J. Phys. Chem.71, 1343 (1967).
Tokiwa, F., J. Phys. Chem.72, 1214 (1968).
Elworthy, P. H. andC. B. Macfarlane, J. Pharm. Pharmacol.17, 65 (1965).
Elworthy, P. H. andC. B. Macfarlane, J. Pharm. Pharmacol.17, 129 (1965).
Attwood, D., J. Phys. Chem.72, 339 (1968).
Elworthy, P. H. andC. B. Macfarlane, J. Chem. Soc. 907 (1963).
Barr, T., J. Oliver, andW. V. Stubbings, J. Soc. Chem. Ind.67, 45 (1948).
Bauer, N. andS. Z. Lewin, inWeissberger, Physical Methods of Organic Chemistry, 3rd Ed., Vol. 1, part 1, p. 153 (New York 1959).
Bauer, N., inWeissberger, Physical Methods of Organic Chemistry, 3rd Ed., Vol. 1, part II, p. 1259 (New York 1959).
Emerson, M. F. andA. Holtzer, J. Phys. Chem.71, 1898 (1967).
Prins, W. andJ. J. Hermans, Koninkl. Ned. Akad. Wetenschap. Proc. B.59, 162 (1956).
Mysels, K. J., J. Colloid Sci.10, 507 (1955);Princen, L. H. andK. J. Mysels, J. Colloid Sci.12, 594 (1957);Mysels, K. J. andL. H. Princen, J. Phys. Chem.63, 1696 (1959).
Parker, R. A. andS. P. Wasik, J. Phys. Chem.62, 967 (1958).
Mukerjee, P., J. Colloid Sci.19, 722 (1964).
Schick, M. J., J. Colloid Sci.17, 801 (1962).
Shinoda, K., Colloidal Surfactants, p. 58 (New York 1963).
Elworthy, P. H. andC. B. Macfarlane, J. Pharm. Pharmacol.14, 100 T (1962).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Attwood, D. The effect of electrolyte on the micellar properties of an anionic-nonionic detergent in aqueous solution. Kolloid-Z.u.Z.Polymere 232, 788–792 (1969). https://doi.org/10.1007/BF01500656
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01500656