Summary
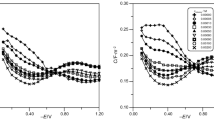
The differential double layer capacities of a dropping mercury electrode in contact with 1 N KF, NaCl, KBr, and KI solutions with dioxane-water mixtures of various volume ratios as solvents were measured by theGrahame ac bridge method. The experimental differential capacity vs. polarization curves showed well-defined suppression by the addition of dioxane over the polarization range near the electrocapillary maximum. While, over the anodic polarization range the increase in the capacity occurred. The theoretical calculation of the diffuse double layer capacity showed that this quantity played a negligible rÔle in the total double layer capacity. Hence, the above phenomena were entirely ascribed to the change in the structure of theStern layer due to the adsorption of dioxane under the present experimental condition, i. e. in the presence of electrolytes at a high concentration.
With increasing polarization in the positive or negative direction, the desorption of dioxane took place due to the increase in the positive free energy of interaction between the double layer field and dioxane molecules. A theory was derived relating the capacity suppression to the surface charge density of mercury, under the assumption of the monolayer adsorption of dioxane. The agreement between the theory and experiments was sufficiently good to obtain reasonable values of the chemical free energy of adsorption.
Similar content being viewed by others

References
Watanabe, A., F. Tsuji andS. Ueda, Proc. Intern. Congr. Surface Activity3, 94 (1957). Bull. Inst. Chem. Res., Kyoto Univ.33, 91 (1955),34, 1, 65 (1956). J. Electrochem. Soc., Japan (in Japanese),22, 521 (1954).
Grahame, D. C., J. Amer. Chem. Soc.63, 1207 (1941),68, 301 (1946);71, 2975 (1949);76, 4819 (1954);78, 3577 (1956).
Ueda, S., F. Tsuji, andA. Watanabe, Bull. Inst. Chem. Res., Kyoto Univ.38, 59 (1960).
Watanabe, A., F. Tsuji, andS. Ueda, unpublished data.
Grahame, D. C., Chem. Revs.41, 441 (1947).
Gouy, L., J. Phys.9, (No. 4) 457 (1910). Ann. Phys.7, (No. 9) 1020, 129 (1917).D. L. Chapman, Phil. Mag.25 (No. 6) 475 (1913).
Devanathan, M. A. V., Trans. Faraday Soc.50, 373 (1954).
Grahame, D. C., J. Chem. Phys.18, 903 (1950).F. Booth, ibid. J. Chem. Phys.19, 391 1327, 1651 (1951).B. E. Conway, J. O'M Bockris, andL. A. Ammar, Trans. Faraday Soc.47, 756 (1951).
Stern, O., Z. Elektrochem.30, 508 (1924).
Devanathan, M. A. V., Proc. Royal Soc.,A 264, 133 (1961).
Debye, P., Z. physik. Chem.130, 55 (1927).
Harned, H. S. andB. B. Owen, The Physical Chemistry of Electrolytic Solutions, 3rd ed., p. 83 (New York 1957).
Weston, L. G., Tables of Electric Dipole Moments, p. 21 (New York 1948).
Guggenheim, E. A., J. physic. Chem.33, 842 (1929).
Author information
Authors and Affiliations
Additional information
The authors wish to express their gratitude to ProfessorI. Tachi, Kyoto University, for his continuous interest and encouragement. Our thanks are also due to ProfessorT. Imaki, Ikenobo Gakuen University, for the purification of dioxane and to Dr.R. Takahashi, Ritsumeikan University, for the polarographic determination of its purity.
Rights and permissions
About this article
Cite this article
Watanabe, A., Tsuji, F. & Ueda, S. Studies on the electrical double layer. Kolloid-Z.u.Z.Polymere 191, 147–153 (1963). https://doi.org/10.1007/BF01499542
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01499542