Summary
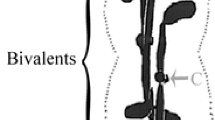
Previous observations indicate that division of the generative cell inTradescantia virginiana is characterized by several unusual features, including persistence of surrounding microtubule (Mt) bundles during karyokinesis, lack of a distinct metaphase plate and direct contribution by mitotic Mts to the cytoskeleton of young sperm. We have further probed karyokinesis in these cells using additional antitubulin and chromosome staining, as well as kinetochore visualizations with CREST serum. The CREST antibodies reveal kinetochores as paired and single fluorescent dots similar to those seen in other species stained with this preparation. Double localizations show that the dots are located at the ends of Mt bundles previously identified as kinetochore fibers (Palevitz and Cresti 1989). Before anaphase, paired kinetochores are distributed along the length of the cell. They also tend to be located at the cell periphery or are directly connected to peripheral Mt bundles by their kinetochore (K)-fibers. Twelve pairs of dots can be counted per cell, equal to the expected number of chromosomes. During anaphase, kinetochore separation starts at various positions along the length of the cell, producing single, relatively uniformly distributed kinetochores in the crotches of forks formed by K-fiber trunks and elongating Mt branches attached to the base of the trunks. Eventually, K-fibers with attached kinetochores aggregate in stepwise fashion on thick Mt bundles at both ends of the cell. This pattern is reflected in the cytoskeleton of young sperm. These results further document the unusual distribution of chromosomes and kinetochores inTradescantia generative cells and the origin of the Mt cytoskeleton in sperm cells.
Similar content being viewed by others
Abbreviations
- CREST:
-
Calcinosis, Raynaud's phenomenon, Esophageal dysmotility, Sclerodactyly, Telangiectasia
- K-fiber:
-
kinetochore fiber
- Mt:
-
microtubule
References
Anderson E, Sax K (1936) A cytological monograph of the American species ofTradescantia. Bot Gaz 97: 433–476
Bajer AS (1968) Behavior and fine structure of spindle fibers during mitosis in endosperm. Chromosoma 25: 249–281
Brenner S, Pepper D, Berns MW, Tan E, Brinkley BR (1981) Kinetochore structure, duplication, and distribution in mammalian cells: analysis by human autoantibodies from scleroderma patients. J Cell Biol 91: 95–102
Brewbaker JL (1967) The distribution and phylogenetic significance of binucleate and trinucleate pollen grains in the angiosperms. Amer J Bot 54: 1069–1083
Brinkley BR, Zinkowski RP, Mollon WL, Davis FM, Pisegna MA, Pershouse M, Rao PN (1988) Movement and segregation of kinetochores experimentally detached from mammalian chromosomes. Nature 336: 251–254
Cassimeris LU, Walker RA, Pryer NK, Salmon ED (1988) Dynamic instability of microtubules. BioEssays 7: 149–154
Charzynska M, Ciampolini F, Cresti M (1988) Generative cell division and sperm cell formation in barley. Sex Plant Reprod 1: 240–247
Cooper DC (1936) Development of the male gametes ofLilium. Bot Gaz 98: 169–177
Gorbsky GJ, Sammak PJ, Borisy GG (1987) Chromosomes move poleward in anaphase along stationary microtubules that co-ordinately disassemble from their kinetochore ends. J Cell Biol 104: 9–18
Johnston GW (1941) Cytological studies on male gamete formation in certain angiosperms. Amer J Bot 28: 306–319
Lafleur GJ, Gross AE, Mascarenhas JP (1981) Optimization of culture conditions for the formation of sperm cells in pollen tubes ofTradescantia. Gamete Res 4: 35–40
Lewandowska E, Charzynska M (1977)Tradescantia bracteata pollen in vitro: pollen tube development and mitosis. Acta Soc Bot Polon 46: 587–597
Maheshwari P (1950) An introduction to the embryology of angiosperms. McGraw-Hill, New York
Molè-Bajer J, Bajer A, Balczon RD, Zinkowski RP, Brinkley BR (1990) Scleroderma CREST antisera recognize kinetochores of higer plantHaemanthus. Proc Natl Acad Sci USA 87: 3599–3603
O'Mara J (1933) Division of the generative nucleus in the pollen tube ofLilium. Bot Gaz 94: 567–578
Palevitz BA, Cresti M (1988) Microtubule organization in the sperm ofTradescantia virginiana. Protoplasma 146: 28–34
—, Cresti M (1989) Cytoskeletal changes during generative cell division and sperm formation inTradescantia virginiana. Protoplasma 150: 54–71
Raudaskoski M, Anstrom H, Pertila K, Virtanen I, Louhelainen J (1987) Role of the microtubule cytoskeleton in pollen tubes: an immunocytochemical and ultrastructural approach. Biol Cell 61: 177–188
Sax K, O'Mara JG (1941) Mechanism of mitosis in pollen tubes. Bot Gaz 102: 629–636
Swedak JA, Forer A (1988) Sex-chromosome anaphase movements in crane-fly spermatocytes are coordinated: ultraviolet microbeam irradiation of one kinetochore of one sex chromosome blocks the movements of both sex chromosomes. J Cell Sci 88: 441–452
Terasaka O, Niitsu T (1989) Peculiar spindle configuration in the pollen tube revealed by the anti-tubulin immunofluorescence method. Bot Mag (Tokyo) 102: 143–147
Wilson HJ (1968) The fine structure of the kinetochore in meiotic cells ofTradescantia. Planta 78: 379–385
Author information
Authors and Affiliations
Additional information
Dedicated to the memory of Professor Oswald Kiermayer
Rights and permissions
About this article
Cite this article
Palevitz, B.A. Kinetochore behavior during generative cell division inTradescantia virginiana . Protoplasma 157, 120–127 (1990). https://doi.org/10.1007/BF01322644
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01322644