Summary
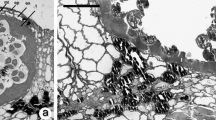
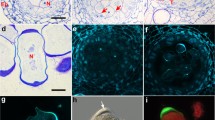
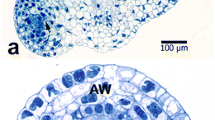
In conifer pollen, the generative cell divides into a sterile stalk cell and a body cell, which subsequently divides to produce two sperm. InPicea abies (Norway spruce, Pinaceae) this spermatogenous body cell contains actin microfilaments. Microfilament bundles follow the spherical contour of the body cell within the cell cortex, and also traverse the cytoplasm and enmesh amyloplasts and other organelles. In addition, microfilaments are associated with the surface of the body cell nucleus. The sterile stalk cell also contains microfilament bundles in the cytoplasm, around organelles, and along the nuclear surface. Within the pollen grain, microfilament bundles traverse the vegetative-cell cytoplasm and are enriched in a webbed cage which surrounds the body cell. Microfilaments were identified with rhodamine-phalloidin and with indirect immunofluo-rescence using a monoclonal antibody to actin. The majority of evidence in literature suggests that the spermatogenous generative cell in angiosperms does not contain actin microfilaments, so the presence of microfilaments within the spermatogenous body cell inP. abies appears to be a fundamental difference in sexual reproduction between conifers and angiosperms.
Similar content being viewed by others
References
Åström H, Virtanen I, Raudaskoski M (1991) Cold stability in the pollen tube cytoskeleton. Protoplasma 161: 99–107
Bohdanowicz J, Ciampolini F, Cresti M (1995) Striped projections of the outer membrane of the generative cell inConvallaria majalis pollen. Sex Plant Reprod 8: 223–227
Brown RC, Lemmon BE (1991a) Pollen development in orchids 3: a novel generative pole microtubule system predicts unequal pollen mitosis. J Cell Sci 99: 273–281
— — (1991b) Pollen development in orchids 5: a generative cell domain involved in spatial control of the hemispherical cell plate. J Cell Sci 100: 559–565
— — (1992) Pollen development in orchids 4: cytoskeleton and ultrastructure of the unequal pollen mitosis inPhalaenopsis. Protoplasma 167: 183–192
Christiansen H (1972) On the development of pollen and the fertilization mechanism ofPicea abies (L.) Karst. Silvae Genet 21: 51–61
Corriveau JL, Coleman AW (1988) Rapid screening method to detect potential biparental inheritance of plastid DNA and results for over 200 angiosperm species. Am J Bot 75: 1443–1458
Dawkins MD, Owens JN (1993) In vitro and in vivo pollen hydration, germination, and pollen tube growth in white spruce,Picea glauca (Moench) Voss. Int J Plant Sci 154: 506–521
de Win AHN, Knuiman B, Pierson ES, Geurts H, Kengen HMP, Derksen J (1996) Development and cellular organization ofPinus sylvestris pollen tubes. Sex Plant Reprod 9: 93–101
Dinis AM, Mesquita JF (1993) The f-actin distribution during microsporogenesis inMagnolia soulangeana Soul. (Magnoliaceae). Sex Plant Reprod 6: 57–63
Frankis RC, Grayson GK (1990) Heat-shock response in germinating pine pollen. Sex Plant Reprod 3: 195–199
Hause G, Hause B, Van Lammeren AAM (1992) Microtubular and actin filament configurations during microspore and pollen development inBrassica napus cv. Topas. Can J Bot 70: 1369–1376
Heslop-Harrison J, Heslop-Harrison Y (1989) Myosin associated with the surfaces of organelles, vegetative nuclei and generative cells in angiosperm pollen grains and tubes. J Cell Sci 94: 319–325
— — (1992a) Cyclical transformations of the actin cytoskeleton of hyacinth pollen subjected to recurrent vapor-phase hydration and dehydration. Biol Cell 75: 245–252
— — (1992b) Intracellular motility, the actin cytoskeleton and germinability in the pollen of wheat (Triticum aestivum L.). Sex Plant Reprod 5: 247–255
— —, Cresti M, Tiezzi A, Ciampolini F (1986) Actin during pollen germination. J Cell Sci 86: 1–8
Hess MW, Mittermann I, Lusctmig C, Valenta R (1995) Immuno-cytochemical localization of actin and profilin in the generative cell of angiosperm pollen: TEM studies on high-pressure frozen and freeze-substitutedLedebouria socialis Roth (Hyacinthaceae). Histochem Cell Biol 104: 443–451
Knox RB, Zee SY, Blomstedt C, Singh MB (1993) Male gametes and fertilization in angiosperms. New Phytol 125: 679–694
Kohno T, Chaen S, Shimmen T (1990) Characterization of the translocator associated with pollen tube organelles. Protoplasma 154: 179–183
Lazzaro MD (1996) The actin microfilament network within elongating pollen tubes of the gymnospermPicea abies (Norway spruce). Protoplasma 194: 186–194
Miller DD, Scordilis SP, Hepler PK (1995) Identification and localization of three classes of myosins in pollen tubes ofLilium longiflorum andNicotiana alata. J Cell Sci 108: 2549–2653
Owens JN, Morris SJ (1990) Cytological basis for cytoplasmic inheritance inPseudotsuga menziesii: I. Pollen tube and archegonial development. Am J Bot 77: 433–445
Palevitz BA, Liu B (1992) Microfilaments (F-actin) in generative cells and sperm: an evaluation. Sex Plant Reprod 5: 89–100
—, Tiezzi A (1992) Organization, composition, and function of the generative cell and sperm cytoskeleton. Int Rev Cytol 140: 149–185
Pettitt JM (1985) Pollen tube development and characteristics of the protein emission in conifers. Ann Bot 56: 379–397
Pierson ES (1988) Rhodamine-phalloidin staining of F-actin in pollen after dimethylsulphoxide permeabilization. Sex Plant Reprod 1: 83–87
—, Cresti M (1992) Cytoskeleton and cytoplasmic organization of pollen and pollen tubes. Int Rev Cytol 140: 73–125
Russell SD (1996) Attraction and transport of male gametes for fertilization. Sex Plant Reprod 9: 337–342
Singh H (1978) Embryology of gymnosperms. Gebrüder Borntraeger, Berlin [Braun HJ et al (eds) Handbuch der Pflanzenanatomie, vol 10, part 2]
Sorri O, Åström H, Raudaskoski M (1996) Actin and tubulin expression and isotype pattern during tobacco pollen tube growth. Sex Plant Reprod 9: 255–263
Tang X, Hepler PK, Scordilis SP (1989a) Immunochemical and immunocytochemical identification of a myosin heavy chain polypeptide inNicotiana pollen tubes. J Cell Sci 92: 569–574
—, Lancelle SA, Hepler PK (1989b) Fluorescence microscopic localization of actin in pollen tubes: comparison of actin antibody and phalloidin staining. Cell Motil Cytoskeleton 12: 216–224
Taylor P, Kenrick J, Li Y, Kaul V, Gunning BES, Knox RB (1989) The male germ unit ofRhododendron: quantitative cytology, three-dimensional reconstruction, isolation and detection using fluorescent probes. Sex Plant Reprod 2: 254–264
Terasaka O, Niitsu T (1994) Differential roles of microtubule and actin-myosin cytoskeleton in the growth ofPinus pollen tubes. Sex Plant Reprod 7: 264–272
Tirlapur UK, Cai G, Faleri C, Moscatelli A, Scali M, Del Casino C, Tiezzi A, Cresti M (1995) Confocal imaging and immunogold electron microscopy of changes in distribution of myosin during pollen hydration, germination and pollen tube growth inNicotiana tabacum L. Eur J Cell Biol 67: 209–217
—, Faleri C, Cresti M (1996) Immunoelectron microscopy of myosin associated with the generative cell in pollen tubes ofNicotiana tabacum L. Sex Plant Reprod 9: 233–237
Tiwari SC, Polito VS (1988) Organization of the cytoskeleton in pollen tubes ofPyrus communis: a study employing conventional and freeze-substitution electron microscopy, immunofluorescence, and rhodamine-phalloidin. Protoplasma 147: 100–112
— — (1990) An analysis of the role of actin during pollen activation leading to germination in pear (Pyrus communis L.): treatment with cytochalasin D. Sex Plant Reprod 3: 121–129
Wick SM (1993) Immunolabeling of antigens in plant cells. Methods Cell Biol 37: 171–200
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lazzaro, M.D. The spermatogenous body cell of the coniferPicea abies (Norway spruce) contains actin microfilaments. Protoplasma 201, 194–201 (1998). https://doi.org/10.1007/BF01287415
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01287415