Summary
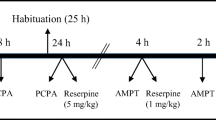
Forskolin is a diterpene derivative that activates adenylate cyclase and raises cAMP levels in the cell. Both ip and icv forskolin cause behavioral hypoactivity. Lithium has been reported for many years to block cAMP accumulation, but the behavioral relevance of this biochemical effect is not clear. We studied the effect of acute and chronic lithium on icv forskolininduced hypoactivity. Acute lithium had no effect, but chronic lithium significantly blocked forskolin-induced hypoactivity. The effect of chronic lithium occurred with both forskolin in DMSO and with a water-soluble forskolin derivative. These results suggest that this behavioral model can be used to investigate whether new inhibitors of adenylate cyclase possess lithium-like effects.
Similar content being viewed by others
References
Andersen PH, Geisler A (1984) Lithium inhibition of forskolin-stimulated adenylate cyclase. Neuropsychobiology 12: 1–3
Avissar S, Schreiber G, Danon A, Belmaker RH (1988) Lithium inhibits adrenergic and cholinergic increases in GTP binding in rat cortex. Nature 331: 440–442
Barraco RA, Phillis JW, Altman HJ (1985) Depressant effect of forskolin on spontaneous locomotor activity in mice. Gen Pharmacol 16: 521–524
Belmaker RH (1981) Receptors, adenylate cyclase, depression and lithium. Biol Psychiatry 16: 333–350
Belmaker RH, Roitman G (1988) A clinical trial of demethylchlortetracycline as a lithium-like agent in excited psychoses. In: Birch NJ (ed) Lithium: inorganic pharmacology and psychiatric use. IRL Press, Eynsham Oxford, pp 191–194
Belmaker RH, Zohar J, Ebstein RP (1980) Cyclic nucleotides in mental disorder. Adv Cycl Nucl Res 12: 187–198
Bender JL, Neer EJ (1983) Properties of the adenylate cyclase catalytic unit from caudate nucleus. J Biol Chem 258: 2432–2439
Bersudsky Y, Kotler M, Shafrin M, Belmaker RH (1996) A preliminary study of possible psychoactive effects of intravenous forskolin in depressed and schizophrenic patients. J Neural Transm 103: 1463–1467
Cooper DMF, Mons N, Karpen JW (1995) Adenylyl cyclases and the interaction between calcium and cAMP signaling. Nature 374: 421–424
Dooley DJ, Mogilnicka E, Delini-Stula A, Waechter F, Truog A, Wood J (1983) Functional supersensitivity to adrenergic agonists in the rat after DSP-4, a selective noradrenergic neurotoxin. Psychopharmacology 81: 1–5
Ebstein RP, Belmaker RH, Grunhaus L, Rimon R (1976) Lithium inhibition of adrenaline-sensitive adenylate cyclase in humans. Nature 259: 411–413
Ebstein RP, Hermoni M, Belmaker RH (1980) The effect of lithium on noradrenalineinduced cyclic AMP accumulation in rat brain: inhibition after chronic treatment and absence of super-sensitivity. J Pharmacol Exp Ther 213: 161–167
Frazer A, Haugaard ES, Mandels J, Jaugaard N (1975) Effects of intracellular lithium on epinephrine-induced accumulation of cyclic AMP in skeleton muscle. Biochem Pharmacol 24: 2273–2277
Givant Y, Zohar J, Lichtenberg P, Kofman O, Belmaker RH (1990) Chronic lithium attenuates clenbuterol-induced hypoactivity. Lithium 1: 183–185
Goldberg H, Clayman P, Skorecki K (1988) Mechanism of Li inhibition of vasopressinsensitive adenylate cyclase in cultured renal epithelial cells. Am J Physiol 255: F995-F1002
Gross G, Dodt C, Hanft G (1988) Effect of chronic lithium administration on adrenoceptor binding and adrenoceptor regulation in rat cerebral cortex. Naunyn Schmiedebergs Arch Pharmacol 337: 267–272
Hamburger-Bar R, Robert M, Newman M, Belmaker RH (1986) Interstrain correlation between behavioral effects of lithium and effects on cortical cyclic AMP. Pharmacol Biochem Behav 24: 9–13
Horowski R, Sastre-Y-Hernandez M (1985) Clinical effects of neurotropic selective cAMP phosphodiesterase inhibitor rolipram in depressed patients: global evaluation of the preliminary reports. Curr Ther Res 38: 23–29
Kaplan Z, Schrieber G, Belmaker RH (1988) Lack of effectiveness of forskolin or inositol as antidote in lithium toxicity. J Neural Transm 72: 167–170
Kofman O, Belmaker RH (1993) Biochemical, behavioral and clinical studies of the role of inositol in lithium treatment and depression. Biol Psychiatry 34: 839–852
Kofman O, Klein E, Newman M, Hamburger R, Kimche O, Nir T, Shimon H, Belmaker RH (1990) Inhibition by antibiotic tetracyclines of rat cortical noradrenergic adenylate cyclase and amphetamine-induced hyperactivity. Pharmacol Biochem Behav 37: 417–424
Kofman O, Belmaker RH, Mork A, Geisler A (1994) The 4-dedimethylamino derivative of tetracycline loses both ability to block cyclic AMP accumulation and the ability to inhibit rat motor activity. Eur Neuropsychopharmacol 4: 420–421
Lerer B, Globus M, Brik E, Hamburger R, Belmaker RH (1984) Effect of treatment and withdrawal from chronic lithium in rats on stimulant-induced responses. Neuropsychobiology 11: 28–32
Marone G, Petracca R, Vigorita S, Casolaro V (1990) Adenosine receptors of human leukocytes. II. Characterization of an inhibitory P-site. Biochem Pharmacol 40: 1963–1973
Mattia A, El-Fakahany EE, Moreton JE (1986) Behavioral and receptor binding studies of phencyclidine (PCP) and lithium interaction in rat. Life Sci 38: 975–984
Mons N, Harry A, Dubourg P, Premont RT, Iyengar R, Cooper DMF (1995) Immunohistochemical localization of adenylyl cyclase in rat brain indicates a highly selective concentration at synapses. Proc Natl Acad Sci USA 92: 8473–8477
Morita T, Wheeler MA, Weiss RM (1986) Relaxant effect of forskolin in rabbit detrusor smooth muscle: role of cyclic AMP. J Urol 135: 1293–1295
Mørk A, Geisler A (1987a) Effects of lithium on calmodulin-stimulated adenylate cyclase activity in cortical membranes from rat brain. Pharmacol Toxicol 60: 17–23
Mørk A, Geisler A (1987b) Mode of action of lithium on the catalytic unit of adenylate cyclase from rat brain. Pharmacol Toxicol 60: 241–248
Mørk A, Geisler A (1994) Lithium in situ decreases extracellular levels of cyclic AMP in the dorsal hippocampus of living rats. Pharmacol Toxicol 74: 300–302
Mørk A, Geisler A (1995) Effect of chronic lithium treatment on agonist-enhanced extracellular concentrations of cyclic AMP in the dorsal hippocampus of freely moving rats. J Neurochem 65: 134–139
Newman ME, Belmaker RH (1987) Effects of lithium in vitro and ex vivo on components of the adenylate cyclase system in membranes from cerebral cortex of the rat. Neuropharmacology 26: 211–217
Overstreet DH, Double K, Schiller GD (1989) Antidepressant effects of rolipram in a genetic animal model of depression: cholinegic supersensitivity and weight gain. Pharmacol Biochem Behav 34: 691–696
Seamon KB, Padget W, Daly JW (1981) Forskolin: unique diterpene activator of adenylate cyclase in the membranes and in intact cells. Proc Natl Acad Sci USA 78: 3363–3367
Skolnick P, Daly JW (1974) Norepinephrine-sensitive adenylate cyclases in rat brains: relation to behavior and tyrosine hydroxylase. Science 184: 174–176
Smith DE (1990) Effects of lithium and rolipram enantiomers on locomotor activity in inbred mice. Pharmacol Toxicol 66: 142–145
Wachtel H (1982) Characteristic behavioral alterations in rats induced by rolipram and other selective adenosine cyclic 3′,5′-monophosphate phosphodiesterase inhibitors. Psychopharmacology 77: 309–316
Wachtel H (1989) Dysbalance of neural second messenger function in the aetiology of affective disorders: a pathophysiological concept hypothesising defects beyond first messenger receptors. J Neural Transm 75: 21–29
Wachtel H, Löschmann P-A, Schneider HH, Rettig K-J (1987) Effects of forskolin on spontaneous behavior, rectal temperature and brain cAMP levels of rats: interaction with rolipram. Neurosci Lett 76: 191–196
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bersudsky, Y., Patishi, Y., Bitsch Jensen, J. et al. The effect of acute and chronic lithium on forskolin-induced reduction of rat activity. J. Neural Transmission 104, 943–952 (1997). https://doi.org/10.1007/BF01285562
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01285562