Summary
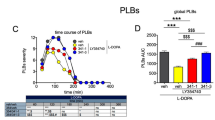
Four experiments were performed to investigate the anti-akinesia effects of combining a sub-threshold dose (5mg/kg, s.c.) of L-Dopa with different doses and combinations of COMT and MAO inhibitors upon the hypokinesia observed in MPTP-treated mice. Ro 40-7592 (1 and 3 mg/kg, s.c.), a novel COMT inhibitor, 60 min before L-Dopa reinstated both locomotion and rearing during a 2-hr interval after L-Dopa in MPTP mice; control mice were unaffected. The combination of Ro 40-7592 (3 mg/kg, s.c.) and pargyline (5 mg/kg, s.c.), a MAO inhibitor, with L-Dopa produced increases in both the peak effect and duration of action indicating a distinct potentiation of the effects of Ro 40-7592 by pargyline. L-Deprenyl, a MAOB inhibitor, together with L-Dopa, restored locomotion and rearing behaviour at all three doses applied (1, 3 and 10 mg/kg, s.c.); in control mice, motor activity was stimulated at the higher doses (3 and 10 mg/kg, s.c.), independent of L-Dopa administration. Combining L-Deprenyl (3 mg/kg, s.c.) with Ro 40-7592 (3 mg/kg, s.c.) one hr before L-Dopa to MPTP mice potentiated the restorative effects of each compound by itself, although no increase in peak effect was obtained. In the control mice, L-Deprenyl plus Ro 40-7592 or L-Deprenyl, by itself, stimulated motor activity following injection of L-Dopa. Marked dopamine (DA) depletions in the striatum of MPTP-treated mice were evident. The present results demonstrate that the effects of the COMT/MAO inhibitors in combination, and in conjunction with L-Dopa (at a dose that was without effect by itself), were well in excess of a summation of their individual effects. It was concluded therefore that a synergism of the restorative, anti-akinesic action of these compounds in MPTP-treated mice could offer a broader therapeutic spectrum in the treatment of Parkinson's disorder.
Similar content being viewed by others
References
Acquas E, Carboni E, de Ree RH, Da Prada M, Di Chiara G (1992) Extracellular concentrations of dopamine and metabolites in the rat caudate after oral administration of a novel catechol-O-methyltransferase inhibitor Ro 40-7592. J Neurochem 59: 326–330
Archer T, Fredriksson A, Jonsson G, Lewander T, Mohammed AK, Ross SB, Söderberg U (1986) Central noradrenaline depletion antagonizes aspects of d-amphetamineinduced hyperactivity in the rat. Psychopharmacology (Berl) 88: 141–146
Barbeau A (1969) L-Dopa therapy in Parkinson's disease: a critical review of nine year's experience. Can Med Assoc J 101: 59–68
Barbeau A (1971) Long-term side-effects of levodopa. Lancet 1: 395
Birkmayer W, Knoll J, Riederer P, Youdim MBH (1983) (−) Deprenyl leads to prolongation of L-DOPA efficacy in Parkinson's disease. Mod Probl Pharmacopsychiatry 19: 170–176
Birkmayer W, Riederer PM, Hars V, Marton J (1985) Improvement of life expectancy due to L-Deprenyl addition to Madopar treatment in Parkinson's disease: a longterm study. J Neural Transm 64: 113–127
Birkmayer W, Riederer PM, Youdim MBH, Linauer W (1975) The potentiation of the anti-kinetic effect after L-DOPA treatment by an inhibitor of MAO-B, deprenyl. J Neural Transm 36: 303–326
Brannan T, Martinez-Tica J, Yahr MD (1992) Catechol-O-methyltransferase inhibition increases striatal L-dopa and dopamine: an in vivo study in rats. Neurology 42: 683–685
Cedarbaum JM, Leger G, Guttman M (1991) Reduction of circulating 3-O-methyltdopa by inhibition of catechol-O-methyltransferase with OR-611 and OR-462 in cynomolgus monkeys: implications for the treatment of Parkinson's disease. Clin Neuropharmacol 14: 330–342
Cedarbaum JM, Leger G, Reches A, Guttman M (1990) Effect of nitecapone (OR-462) on the pharmacokinetics of levodopa and 3-O-methyldopa formation in cynomolgus monkeys. Clin Neuropharmacol 13: 544–552
Chrisp P, Mammen GJ, Sorkin EM (1991) Selegiline. A review of its pharmacology, symptomatic benefits and protective potential in Parkinson's disease. Drugs Aging 1: 228–248
Da Prada M (1991) New approaches to the treatment of age-related brain disorders. Can J Neurol Sci 18: 384–386
Davis GC, Williams AC, Markey SP, Ebert MH, Caine ED, Reichert CM, Kopin IJ (1979) Chronic parkinsonism secondary to intravenous injection of meperidine analogues. Psychiatry Res 1: 249–254
Drago F, Continella G, Spadaro F, Cavaliere S, Scapagnini U (1986) Behavioral effects of deprenyl in aged rats. Funct Neurol 1: 165–174
Engberg G, Elebring T, Nissbrandt H (1991) Deprenyl (selegiline), a selective MAO-B inhibitor with active metabolites; effects on locomotor activity, dopaminergic neurotransmission and firing rate of nigral dopamine neurons. J Pharmacol Exp Ther 259: 841–847
Fahn S, Comi R, Snider SR, Prasad ALN (1979) Effect of a catechol-O-methyl transferase inhibitor, U-0521; with levodopa administration. Biochem Pharmacol 28: 1221–1225
Forna KS (1982) Pathology of Parkinson's disease. In: Marsden CD, Fahn S (eds) Movement disorders. Butterworth Scientific, London, pp 25–40
Fredriksson A, Archer T (1994) MPTP-induced behavioural and biochemical deficits: a paramatric analysis. J Neural Transm 7: 123–132
Fredriksson A, Plaznik A, Sundström E, Jonsson G, Archer T (1990) MPTP-induced hypoactivity in mice: reversal by L-dopa. Pharmacol Toxicol 67: 295–301
Gervas J, Muradas B, Bazan E, Aquado E, de Yebenes J (1983) Effects of 3-OM-dopa on monoamine metabolism in rat brain. Neurology 33: 278–282
Goldstein M, Lieberman A (1992) The role of the regulatory enzymes of catecholamine synthesis in Parkinson's disease. Neurology 42: 8–12
Guttman M, Leger G, Reches A, Evans A, Kuwabara H, Cedarbaum JM, Gjedde A (1993) Administration of the new COMT inhibitor OR-611 increases striatal uptake of fluorodopa. Mov Disord 8: 298–304
Heikkila RE, Cabbat FS, Manzino L, Duvoisin RC (1981) Potentiation by deprenil of L-dopa induced circling in nigral lesioned rats. Pharmacol Biochem Behav 15: 75–79
Heikkila RE, Sieber B-A, Manzino L, Sonsalla PK (1989) Some features of the nigrostriatal dopaminergic neurotoxin 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) in the mouse. Mol Chem Neuropathol 10: 171–183
Heinonen EH, Lammintausta R (1991) A review of the pharmacology of selegiline. Acta Neurol Scand [Suppl] 136: 44–59
Heinonen EH, Myllylä V, Sotaniemi K, Lamintausta R, Salonen JS, Anttila M, Savijärvi M, Kotila M, Rinne UK (1989) Pharmacokinetics and metabolism of selegiline. Acta Neurol Scand 80 [Suppl 126]: 83–91
Hietanen MH (1991) Selegiline and cognitive function in Parkinson's disease. Acta Neurol Scand 84: 407–410
Jonsson G, Hallman H, Mefford I, Adams RN (1980) The use of liquid chromamatography with electrochemical detection for the determination of adrenaline and other biogenic amines in the CNS. In: Fuxe K, Goldstein M, Hökfelt B, Hökfelt T (eds) Central adrenaline neurones. Pergamon Press, Oxford, pp 59–71
Kaakkola S, Wurtman RJ (1992) Effects of COMT inhibitors on striatal dopamine metabolism: a microdialysis study. Brain Res 587: 241–249
Kaakkola S, Teravainen H, Ahtila S, Rita H, Gordin A (1994) Effect of entacapone, a COMT inhibitor, on clinical disability and L-Dopa metabolism in parkinsonian patients. Neurology 44: 77–80
Karoum F, Chuang L-W, Eisler T, Calne DB, Liebowitz MR, Quitkin FM, Klein DF, Wyatt RJ (1982) Metabolism of (−) deprenyl to amphetamine and methamphetamine may be responsible for deprenyl's therapeutic benefit: a biological assessment. Neurology 32: 503–509
Keller R, Oke A, Mefford I, Adams RN (1976) Liquid chromatographic analysis of the catecholamines — routine assay for regional brain mapping. Life Sci 19: 995–1004
Keranen T, Gordin A, Harjola VP, Karlsson M, Korpela K, Pentikainen PJ, Rita H, Seppala L, Wikberg T (1993) The effect of catechol-O-methyl transferase inhibition by entacapone on the pharmacokinetics and metabolism of levodopa in healthy volunteers. Clin Neuropharmacol 16: 145–156
Kirk RE (1968) Experimental design. Procedures in behavioural science. Belmont CA, Brooks/Cole Inc
Knoll J (1978) The possible mechanisms of action of (−) deprenyl in Parkinson's disease. J Neural Transm 43: 177–198
Knoll J (1987) (−) Deprenyl (selegiline, MoverganR) facilitates the activity of the nigrostriatal neuron. J Neural Transm [Suppl 25]: 45–66
Knoll J (1989) The pharmacology of selegiline [(−) deprenyl]. New aspects. Acta Neurol Scand 80 [Suppl 126]: 83–91
Knoll J (1992) Pharmacological basis of the therapeutic effect of (−) deprenyl in agerelated neurological diseases. Med Res Rev 12: 505–524
Laihinen A, Rinne JO, Rinne UK, Haaparanta M, Ruotsalainen U, Bergman J, Solin O (1992) [18F]-6-fluorodopa PET scanning in Parkinson's disease after selective COMT inhibition with nitecapone (OR-462). Neurology 42: 199–203
Langsten JW, Ballard P, Tetrud JW, Irwin I (1983) Chronoic parkinsonism in humans due to a product of meperidine analog. Science 219: 979–980
Lees AJ, Shaw KM, Kohout LJ, Stern GM, Elsworth JD, Sandler M, Youdim MBH (1977) Deprenyl in Parkinson's disease. Lancet 2: 791–796
LeWitt PA (1991) Deprenyl's effect at slowing progression of parkinsonian disability: the DATATOP study. The Parkinson Study Group. Acta Neurol Scand [Suppl 136]: 79–86
Limousin P, Pollak P, Gervason-Tournier CL, Hommel M, Perret JE (1993) Ro 40-7592, a COMT inhibitor, plus levodopa in Parkinson's disease. Lancet 341: 1605
Lindén I-B, Etemadzedah E, Schultz E, Pohto P (1990) Selective catachol-O-methyltransferase inhibition as potential adjunctive treatment with L-dopa in Parkinson's disease. Mov Disord 5: 49
Maj J, Rogoz Z, Skuza G, Sowinska H, Superata J (1990) Behavioural and neurochemical effects of Ro 40-7592, a new COMT inhibitor with a potential therapeutic activity in Parkinson's disease. J Neural Transm [P-D Sect] 2: 101–112
Männistö P, Kaakkola S (1990) Rationale for selective COMT inhibitors as adjuncts in the treatment of Parkinson's disease. Pharmacol Toxicol 66: 317–323
Männistö PT, Tuomainen P, Toivonen M, Tornwall M, Kaakkola S (1990) Effect of acute levodopa on brain catecholamines after selective MAO and COMT inhibition in male rats. J Neural Transm [P-D Sect] 2: 31–43
Muenter M, Sharpless N, Tyce G (1972) Plasma 3-O-methyldopa in L-dopa therapy of Parkinson's disease. Mayo Clin Proc 47: 389–395
Nakashima H, Asari S, Nishimoto A, Goldstein M (1991) The effect of chronic treatment of deprenyl in animal models of Parkinson's disease. No To Shinkei 43: 357–361
Nomoto M, Fukuda T (1993) A selective MAO-B inhibitor Ro19-6327 potentiates the effects of levodopa on parkinsonism induced by MPTP in the common marmoset. Neuropharmacology 32: 473–477
Nutt J, Woodward W (1987) 3-O-Methyldopa and the response to levodopa. Ann Neurol 21: 584–588
Nuutila J, Kaakkola S, Männistö PT (1987) Potentiation of central effects of L-dopa by an inhibitor of catechol-O-methyltransferase. J Neural Transm 70: 233–240
Okuda C, Segal DS, Kuczenski P (1992) Deprenyl alters behavior and caduate dopamine through an amphetamine-like action. Pharmacol Biochem Behav 43: 1075–1080
Reches A, Fahn S (1982) 3-O-Methyldopa blocks dopa metabolism in rat corpus striatum. Ann Neurol 12: 267–271
Reches A, Fahn S (1984) Catechol-O-methyltransferase and Parkinson's disease. Adv Neurol 40: 171–178
Reches A, Miekle L, Fahn S (1982) 3-O-Methyldopa inhibits rotations induced by levodopa in rats after unilateral destruction of the nigrostriatal pathway. Neurology 32: 887–888
Reynolds JP, Elsworth JD, Blau K, Sandler M, Lees AJ, Stern GM (1978) Deprenyl is metabolized to methamphetamine and amphetamine in man. Br J Clin Parmacol 6: 543–544
Rinne UK (1989) Combination of a dopamine agonist, MAO-B inhibitor and levodopa — a new strategy in the treatment of early Parkinson's disease. Acta Neurol Scand [Suppl 126]: 165–169
Rinne UK, Surtola T, Sonniven V (1978) L-Deprenyl treatment of on-off phenomena in Parkinson's disease. J Neural Transm 43: 253–262
Selby G (1968) Parkinson's disease. In: Vinken PJ, Bruyn GW (eds) Handbook of clinical neurology. North-Holland, Amsterdam, pp 173–211
Sharpless N, Muenter M, Tyce G (1972) 3-Methoxy-4-hydroxyphenylalanine (3-O-methyldopa) in plasma during oral L-dopa therapy of patients with Parkinson's disease. Clin Chim Acta 37: 359–369
Sonsalla PK, Heikkila RE (1986) The influence of dose and dosing interval on MPTP-induced dopaminergic neurotoxicity in mice. Eur J Pharmacol 129: 339–345
Teravainen H (1990) Selegiline in Parkinson's disease. Acta Neurol Scand 81: 333–336
Tetrud JW, Langston JW (1989) The effect of deprenyl (Selegiline) on the natural history of Parkinson's disease. Science 245: 519–522
Timar J, Knoll B, Knoll J (1992) (−) Deprenyl is devoid of amphetamine-like behavioural effects in rats. Acta Physiol Hung 79: 131–137
Timar J, Gyarmati Z, Tekes K, Harsing GL, Knoll J (1993) Further proof that (−) deprenyl fails to facilitate mesolimbic dopaminergic activity. Pharmacol Biochem Behav 46: 709–714
Walton J (1985) Brain's diseases of the nervous system, 9th ed. Oxford University Press, New York
Weihmüller FB, Hadjiconstantinou M, Bruno JP (1989) Dissociation between the biochemical and behavioural recovery in MPTP-treated mice. Pharmacol Biochem Behav 34: 113–117
Yasar S, Schindler CW, Thorndike EB, Szelenyi I, Goldberg SR (1993) Evaluation of the stereoisomers of deprenyl for amphetamine-like discriminative stimulus effects in rats. J Pharmacol Exp Ther 265: 1–6
Yoshida T, Oguro T, Kuroiwa Y (1987) Hepatic and extrahepatic metabolism of deprenyl, a selective monoamine oxidase (MAO) B inhibitor, of amphetamines in rats: sex and strain differences. Xenobiotica 17: 957–963
Yoshida T, Yamada Y, Yamamoto T, Kuroiwa Y (1986) Metabolism of deprenyl, a selective monoamine oxidase (MAO) B inhibitor in rat: relationship of metabolism to MAO-B inhibitory potency. Xenobiotica 16: 129–136
Zurcher G, Colzi A, Da Prada M (1990) Ro 40-7592: inhibition of COMT in rat brain and extracerebral tissues. J Neural Transm [Suppl 32]: 375–380
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Fredriksson, A., Archer, T. Synergistic interactions between COMT-/MAO-inhibitors and L-Dopa in MPTP-treated mice. J. Neural Transmission 102, 19–34 (1995). https://doi.org/10.1007/BF01276562
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01276562