Abstract
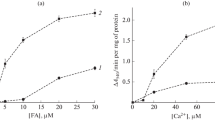
Palmitoyl CoA and palmitoyl carnitine added to rat heart mitochondria in amounts above 20 and 50 nmoles/mg protein, respectively, induced a fall in transmembrane potential and loss of endogenous Mg2+. The dissipation of membrane potential by low concentrations of palmitoyl CoA in the presence of C2+, but not that of high concentrations of palmitoyl CoA alone, was prevented by either ruthenium red, Cyclosporin A or Mg2+, but reversed only by Mg2+. The fall of membrane potential induced by palmitoyl carnitine was not prevented by any of these factors. It is suggested that the action of both palmitoyl CoA and palmitoyl carnitine at high concentrations is due to a non specific disruption of membrane architecture, while that of low concentrations of palmitoyl CoA in the presence of Ca2+ is associated specifically with energy dissipation due to Ca2+ cycling.
Similar content being viewed by others
Abbreviations
- LCACoA:
-
Long-Chain Acyl CoAs
- LCAcar:
-
Long-Chain Acyl carnitines
- Pcar:
-
Palmitoyl carnitine
- PCoA:
-
Palmitoyl CoA
- ΔΨ:
-
Transmembrane Potential
References
Neely JR, Garber D, McDonough K, Idell-Wenger J: Relationship between ventricular function and intermediates of fatty acid metabolism during myocardial ischemia: effects of carnitine. In: MM Winbury and Y Abisko (eds) Ischemic myocardium and antianginal drugs. Raven Press, New York, 1979, pp 225–234
Brecher P: The interaction of long-chain acyl CoA with membranes. Mot Cell Biochem 57: 3–15, 1983
Lamers JMJ, Stinis HT, Montfoort AD, Hulsmann WC: The effect of lipid intermediates on Ca2+ and Na− permeability and (Na+ + K+) ATPase of cardiac sarcolemma. A possible role in myocardial ischemia. Biochim Biophys Acta 774: 127–137, 1984
Pande SV, Blanchaer MC: Reversible inhibition of mitochondrial adenosine diphosphate phosphorylation by long acyl coenzyme A esters. J Biol Chem 246: 402–411, 1971
Morel F, Laquin C, Lunardi J, Duszynski J, Vignais PV: An appraisal of the functional significance of the inhibitory effect of long chain acyl-CoAs on mitochondrial transports. FEBS Lett 39: 133–138, 1974
Devaux PF, Bienvenue A, Lauquin C, Brisson AD, Vignais M, Vignais PV: Interaction between spin-labeled acyl-coenzyme A and the mitochondrial adenosine diphosphate carrier. Biochemistry 14: 1272–1280, 1975
Beatrice MC, Pfeiffer DR: The mechanism of palmitoyl-CoA inhibition of Ca2+ uptake in liver and heart mitochondria. Biochem J 194: 71–77, 1981
Di Lisa F, Menabò R, Miotto G, Bobyleva-Guarriero V, Siliprandi N: Ca2+-mediated action of long-chain acyl-CoA on liver mitochondria energy-linked processes. Biochim Biophys Acta 973: 185–188, 1989
Le Quoc K, Le Quoc D: Involvement of the ADP/ATP carrier in calcium-induced perturbations of the mitochondrial inner membrane permeability: Importance of the orientation of the nucleotide binding site. Arch Biochem Biophys 265: 249–257, 1988
Piper MH, Sezer O, Schwartz P, Hutter JF, Schweikhardt C, Spieckermann PG: Acylcarnitine effects on isolated cardiac mitochondria and erythrocites. Basic Res Cardiol 79: 186–198, 1984
Sordhal LA, Johnson C, Blailock ZR, Schwartz A: The mitochondrion. Methods in Pharmacol 1: 247–283, 1971
Kamo N, Muratsugu M, Hongoh R, Kobatake Y: Membrane potential of mitochondria measured with an electrode sensitive to tetraphenylphosphonium and relationship between proton electrochemical potential and phosphorylation potential in steady state. Membr Biol 49: 105–121, 1979
Affolter H, Siegel E: A simple system for the measurement of ion activities with solvent polymeric membrane electrodes. Anal Biochem 97: 315–319, 1979
Jensen SD, Gunter KK, Gunter TE: The efficiencies of the component steps of oxidative phosphorylation ii. Experimental determination in mitochondria and examination of the equivalence of membrane potential and pH gradient in phosphorylation. Arch Biochem Biophys 248: 305–323, 1986
Toninello A, Siliprandi D, Siliprandi N: Mg2+ restores membrane potential in rat liver mitochondria deenergized by Cal2+ and phosphate movements. FEBS Lett 142: 63–66, 1982
Stucki JW, Ineichen EA: Energy dissipation by calcium recycling and the efficiency of calcium transport in rat liver mitochondria. Eur J Biochem 48: 365–375, 1974
Fournier NC, Richard MA: Fatty acid-binding protein, a potential regulator of energy production in the heart. J Biol Chem 263: 14471–14479, 1988
Crompton M, Ellinger H, Costi A: Inhibition by cyclosporin A of a Ca2+-dependent pore in heart mitochondria activated by inorganic phosphate and oxidative stress. Biochem J 255: 357–360, 1988
Idell-Wenger JA, Grotyohann LW, Neely JR: Coenzyme A and carnitine distribution in normal and ischemic hearts. J Biol Chem 253: 4310–4318, 1978
Zoccarato F, Rugolo M, Siliprandi D, Siliprandi N: Correlated effluxes of adenine nucleotides, Mg2+ and Ca2+ induced in rat liver mitochondria by external Ca2+ and phosphate. Eur J Biochem 114: 195–199, 1981
Toninello A, Siliprandi D, Siliprandi N: On the mechanism by which Mg2+ and adenine nucleotides restore membrane potential in rat liver mitochondria deenergized by Ca2+ and phosphate. Biochim Biophys Res Comm 111: 792–797, 1983
Siliprandi D, Di Lisa F, Toninello A: On the role of Mg2+ in heart mitochondria. In: MC Berman, W Gevers, LH Opie (eds) Membranes and Muscle. Published for the ICSU Press by IRL Press, 1985, pp 378–379
Panov A, Filippova S, Lyakhovich V: Adenine nucleotide translocase as a site of regulation by ADP of the rat liver mitochondria permeability to H+ and K+ ions. Arch Biochem Biophys 199: 420–426, 1980
Griffiths EJ, Halestrap AP: Further evidence that cyclosporin A protects mitochondria from calcium overload by inhibiting a matrix peptidil-prolyl cis-trans isomerase. Biochem J 274: 611–614, 1991
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Siliprandi, D., Biban, C., Testa, S. et al. Effects of palmitoyl CoA and palmitoyl carnitine on the membrane potential and Mg2+ content of rat heart mitochondria. Mol Cell Biochem 116, 117–123 (1992). https://doi.org/10.1007/BF01270578
Issue Date:
DOI: https://doi.org/10.1007/BF01270578