Abstract
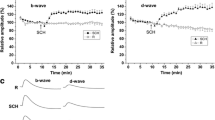
Single-cell electrophysiologic studies have shown that dopamine modulates retinal activity, but its role in human retinal processing is unclear. We investigated the effects of short-term oral administration of dopaminergic receptor blocking agents on the flash electroretinogram in humans. Both chlorpromazine (25 and 50 mg) and fluphenazine (1 and 2 mg) significantly reduced electroretinogram b-wave amplitudes and also selectively reduced the amplitude of the first oscillatory potential. Implicit times were not altered. Metoclopramide (10 and 20 mg) had no effect on any electroretinographic variable. Our study indicates that dopamine receptor blocking agents with both D-1 and D-2 receptor affinities reduce the amplitude of the electroretinogram in humans.
Similar content being viewed by others
Abbreviations
- ANOVA:
-
analysis of variance
- VlogI:
-
voltage versus log intensity
References
Kramer SG. Dopamine: A retinal neurotransmitter, I: Retinal uptake, storage, and light-stimulated release of H3-dopamine in vivo. Invest Ophthalmol Vis Sci 1971; 10: 438–52.
Iuvone PM, Galli CL, Garrison-Gund CK, Neff NH. Light activated tyrosine hydroxylase and increased dopamine synthesis in retinal amacrine neurons. Science 1978; 202: 901–2.
Dubocovich ML, Weiner N. Pharmacological differences between the D-2 autoreceptor and the D-1 dopamine receptor in rabbit retina. Pharmacol Exp Ther 1985; 233: 747–54.
Zarbin MA, Warmsley JK, Palacios JM, Kuhar MJ. Auto-radiographic localization of high affinity GABA, benzodiazepine, dopaminergic, adrenergic and muscarinic cholinergic receptors in the rat, monkey and human retina. Brain Res 1986; 374: 75–92.
Kupenova P, Belcheva S. Effects of haloperidol, methylergometrine and phentolamine on the frog ERG. Experientia 1981; 37: 852–4.
Sato T, Yoneyama T, Kim HK, Suzuki TA. Effect of dopamine and haloperidol on the c-wave and light peak of light-induced retinal responses in chick eye. Doc Ophthalmol 1987; 65: 359–71.
Wioland N, Rudolf G, Bonaventure N. Electrooculographic and electroretinographic study in the chicken after dopamine and haloperidol. Doc Ophthalmol 1990; 75: 175–80.
Jagadeesh JM, Lee HC, Salazar-Bookaman M. Influence of chlorpromazine on the rabbit electroretinogram. Invest Ophthalmol Vis Sci 1980; 19: 1449–56.
Schneider T, Zrenner E. The effect of fluphenazine on rod-mediated retinal responses. Doc Ophthalmol 1987; 65: 287–96.
Oliver P, Jolicoeur FB, Lafond G, Drumheller A, Brunette JR. Effects of retinal dopamine depletion on the rabbit electroretinogram. Doc Ophthalmol 1987; 66: 359–71.
Wong C, Ishibashi T, Tucker G, Hamasaki D. Responses of the pigmented rabbit retina to NMPTP, a chemical inducer of Parkinsonism. Exp Eye Res 1985; 40: 509–19.
Perossini M, Fornaro P. Electroretinographic effects in humans by psychopharmacologic agents. Doc Ophthalmol 1990; 75: 1–6.
Iudice A, Virgili P, Muratorio A. The electroretinogram in Parkinson's disease. Res Commun Psychol Psychiatry Behav 1980; 5: 283–9.
Bartel P, Blom M, Robinson E, van der Meyden C, de Sommers K, Becker P. The effects of levodopa and haloperidol on flash and pattern ERGs and VEPs in normal humans. Doc Ophathalmol 1990; 76: 55–64.
Jaffe MJ, Levinson PD, Zimmlichman R, Coen JC, Karson CN, deMonasterio FM. The effect of metoclopramide on the ganzfeld electroretinogram. Vision Res 1987; 27: 1693–700.
Maruiwa F, Kim S-D, Nao-i N, Sawada A. Effect of metoclopramide, dopamine receptor blocker, on the EOG light peak. Acta Soc Ophthalmol Jpn. 1992; 96: 375–80.
Harnois C, DiPaolo T. Decreased dopamine in the retinas of patients with Parkinson's disease. Invest Ophthalmol Vis Sci 1990; 31: 2473–5.
Ellis CJK, Allen TGJ, Marsden CD, Ikeda H. Electroretinographic abnormalities in idiopathic Parkinson's disease and the effect of levodopa administration. Clin Vision Sci 1987; 1: 347–55.
Gottlob I, Schneider E, Heider W, Skrandies W. Alteration of visual evoked potentials and electroretinograms in Parkinson's disease. Electrophysiol Clin Neurophysiol 1987; 66: 349–57.
Kupersmith MJ, Shakin E, Siegel IM, Lieberman A. Visual system abnormalities in patients with Parkinson's disease. Arch Neurol 1982; 39: 284–6.
Seeman P, Grigoriadis D. Dopamine receptors in brain and periphery. Neurochem Int 1987; 10: 1–25.
Peachey NS, Alexander KR, Fishman GA. Rod and cone system contributions to oscillatory potentials: An explantation for the conditioning flash effect. Vision Res 1987; 27: 859–66.
Hood DC, Birch DG. The a-wave of the human ERG and rod receptor function. Invest Ophthalmol Vis Sci 1990a; 31: 2070–81.
Hood DC, Birch DG. A quantitative measure of the electrical activity of human rod photoreceptors using electroretinography. Vis Neurosci 1990b; 5: 379–84.
Harnois C, Marcotte G, Bedard PJ. Alteration of monkey retinal oscillatory potentials after MPTP injection. Doc Ophthalmol 1988; 67: 363–9.
Marmor MF, Hock P, Schechter G, Pfefferbaum A, Berger PA, Maurice R. Oscillatory potentials as a marker for dopaminergic disease. Doc Ophthalmol 1988; 69: 255–61.
Wachtmeister L, Dowling JE. The oscillatory potentials of the mudpuppy retina. Invest Ophthalmol Vis Sci 1978; 17: 1178–88.
Wachtmeister L. Further studies of the chemical sensitivity of the oscillatory potentials of the electroretinogram (ERG) II: Glutamate-aspartate and dopamine antagonists. Acta Ophthalmol 1981; 59: 247–58.
Jensen RJ, Daw NW. Towards an understanding of the role of dopamine in the mammalian retina. Vision Res 1983; 23: 1293–8.
Ikeda H, Priest TD, Robbins J, Wakakuwa K. Silent dopaminergic synapse at feline retinal ganglion cells? Clin Vision Sci 1986; 1: 25–38.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Holopigian, K., Clewner, L., Seiple, W. et al. The effects of dopamine blockade on the human flash electroretinogram. Doc Ophthalmol 86, 1–10 (1994). https://doi.org/10.1007/BF01224623
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01224623