Abstract
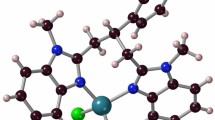
(C22H22CuN4O4Cl2 2·7H2O is triclinic,C 1i− t-P1. Unit cell dimensions at 293 K area=12.028(3),b=20.378(6),c=11.387(3) Å, α=113.10(2),β=84.13(2), γ=95.99(2)°,V=2547.5 0A3,D c =1.573 Mg. m−3, andZ=2. The structure has been determined from single-crystal data collected with a four-circle diffractometer and refined from 3904 reflections down toR=0.054 andR w =0.058. The asymmetric unit is built from two independent A and B molecules in which the copper atom has a square pyramidal environment in A and an octahedral one inB. Intermolecular Cu-O (hydroxymethyl groups) contacts observed in the solid state are partially present in aqueous solutions.
Similar content being viewed by others
References
Busing, W. R., and Levy, H. A. (1962)Orxfls 3. Report ORNL-TM-271 (Oak Ridge National Laboratory, Tennessee).
Busing, W. R., Martin, K. O., and Levy, H. A. (1964)Orffe, Report ORNL-TM-306 (Oak Ridge National Laboratory, Tennessee).
Casella, L., Gullotti, M., and Pacchioni, G. (1982)J. Am, Chem. Soc. 104, 2386–2396, and references cited.
Cassoux, P., and Gleizes, A. (1980)Inorg. Chem. 19, 665–672.
Cromer, D. T. (1965)Acta Cryst. 18, 17–23.
Cromer, D. T., and Waber, J. T. (1974)International Tables for X-ray Crystallography, Vol. IV, Ibers, J. A., and Hamilton, W. C., eds. (Kynoch Press, Birmingham, England), pp. 72–79.
Cutfield, J. F., Hall, J. T., and Waters, T. N. (1967)J. Chem. Soc. Chem. Commun., 785–786.
Davies, J. E., Gatehouse, B. M., and Murray, K. S. (1973)J. Chem. Soc. Dalton. Trans., 2523–2527.
Germain, G., Main, P., and Woolfson, M. M. (1971)Acta Cryst. A 27, 368–376.
Hathaway, B. J., and Billing, D. E. (1970)Coord. Chem. Rev. 5, 143–207.
Hathaway, B. J. (1982)Coord. Chem. Rev. 41, 423–487.
Ikawa, M., and Snell, E. E. (1954)J. Am. Chem. Soc. 76, 4900–4902.
Johnson, C. K. (1965)Ortep, Report ORNL-3794 (Oak Ridge National Laboratory, Tennessee).
Laurent, J. P., Bonnet, J. J., Nepveu, F., Astheimer, H., Walz, L., and Haase, W. (1982)J. Chem. Soc. Dalton Trans., 2433–2438.
Lintvedt, R. L., Tomlonovic, B., Fenton, D. E., and Glick, M. D. (1976),Adv. Chem. Ser. 150, 407–425.
Martell, A. E. (1982)Advances in Enzymology, Vol. 53 (Wiley, New York), pp. 163–199.
McAuliffe, C. A. (1977)J. Chem. Soc. Dalton Trans., 1762–1777.
Mosset, A., Bonnet, J.-J., and Galy, J. (1977)Acta Cryst. B 33, 2639–2644.
Nepveu, F., Bonnet, J.-J., Laurent, J.-P, and Massol, M. (1981)J. Coord. Chem. 11, 185–193.
Nepveu, F., Laurent, J.-P., and Bonnet, J.-J. (1983)Acta Cryst, in press.
Percy, G. C., and Thornton, T. A. (1972)J. Inorg. Nucl. Chem. 34, 3357–3367.
Purdy, E. R. (1976) PhD Thesis (State University of New York at Stony Brook, Diss. Abstr. Int. B37), p. 3406.
Sharrock, P., Nepveu-Juras, F., Massol, M., and Haran, R. (1979)Biochem. Biophys. Res. Commun. 86, 428–433.
Sheldrick, G. (1976)Shelx 76, A program for crystal structure determination (University of Cambridge, England).
Stewart, R. F., Davidson, E. R., and Simpson, W. T. (1965)J. Chem. Phys. 42, 3175–3187.
Varshavsky, Y. S., Cherkasova, T. G., and Buzina, V. A. (1974)J. Organomet. Chem. 77, 117–124.
Ugo, R., La Monica, G., Cenini, S., and Bonati, F. (1968)J. Organomet. Chem. 11, 159–166.
Walz, L., Paulus, H., Haase, W., Langhof, H., and Nepveu, F. (1983)J. Chem. Soc. Dalton Trans., in press.
Yamada, S., and Kuge, K. (1974)Inorg. Chem. Acta 8, 29–32.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nepveu, F., Dahan, F., Haran, R. et al. N,N′-(1,2-phenylene)bis(pyridoxal-hydrochlorideiminato)copper(II): crystallographic and spectroscopic studies. Journal of Crystallographic and Spectroscopic Research 14, 129–142 (1984). https://doi.org/10.1007/BF01189555
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01189555