Summary
-
1.
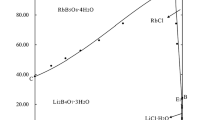
The 25° and 40° solubility isotherms have been determined for the systems LiCl-RbCl-H2O and LiCI -CsCl -H2O.
-
2.
The compounds RbCl - 2LiCl - 4H2O and 3RbCl - LiCl - 2H2O have been found in the first system, and the compound 2CsCl - LiCl - 4H2O has been found in the second system.
Similar content being viewed by others
Literature cited
N. K. Voskresenskaya and O. K. Yanatyeva, Bull. Acad. Sci. USSR Chem. Series, No. 1, 97 (1937).
H. Basset, J. Sandersen, J. Chem. Soc., 1932, 1855.
H. Benraht, Z. anorg. Chem. 220, 145 (1934).
H. Benraht, Z. anorg. Chem. 205, 417 (1932).
F. M. Perelman, Rubidium and Cesium, Acad. Sci. USSR Press, Moscow and Leningrad. 1941.
A. V. Novoselova and I. G. Sosnovskaya, J. Gen. Chem., 21, 813 (1951).
V. P Blidin, Proc. Acad. Sci. USSR, 84, No, 5 (1952).
J. Schreinemakers, Z. Phys. Chem. 11, 81 (1893).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Blidin, V.P. Heterogenrous equilibria in the systems LiCl-RbCI-H2O, and LiCI-CsCI-H2O. Russ Chem Bull 2, 723–726 (1953). https://doi.org/10.1007/BF01178849
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01178849