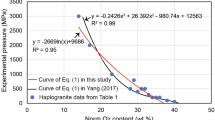
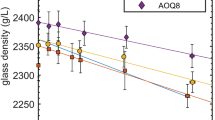
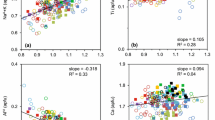
Summary
We present an empirical technique for estimating crystallization pressure and H2O content of MORB and BABB glasses using glass major element compositions alone. This technique is based on models describing 1 atm mineral — basaltic melt equilibria for olivine (ol), plagioclase (pl), and clinopyroxene (cp) which allow calculation of liquidus temperatures for each mineral on the basis of melt composition alone. A necessary requirement for using the proposed technique is cosaturation of glasses with of andpl for estimation of H2O contents, and withol andcp for estimation of crystallization pressures. Mineralogical criteria can be used to assess cosaturation.
The following logic is employed:
Calculated temperatures ofol, pl andcp liquidus must be the same for a glass which represents a quenched melt which crystallized these minerals at 1 atm. The effect ofpl, forcingcp to appear earlier on the liquidus. For higher-pressure crystallization, calculated 1 atm temperature forcp must be lower than that forol due to stronger dependence ofcp crystallization temperature on pressure. Thus the difference between calculated 1 atm liquidus temperatures forol andcp must reflect crystallization pressure. It is well known that H2O can depress crystallization temperature ofpl compared to those ofol andcp. Thus forol-pl saturated H2O-bearing melts, calculated 1 atm anhydrous temperatures forpl must be higher than those forol, and the difference between calculated anhydrous 1 atm liquidus temperatures forpl andol must reflect melt H2O content. p ]As the results of this technique are entirely dependent on the quality of glass major element analyses, we emphasize that application of this method to a single glass analysis can lead to erroneous results. A preferable approach is to obtain a statistically meaningful number of glass analyses for each suite and use averages. We also emphasize the danger of extrapolation of our technique to compositions beyond the MORB — BABB compositional range.
Zusammenfassung
Wir präsentieren eine empirische Methode zur Abschätzung des Kristallisationsdruckes und des H2O-Gehaltes von MORB und BABB Gläsern, wobei auschließlich die Hauptelementzusammensetzung der Gläser benützt wird. Diese Methode beruht auf Modellen, die die Schmelzgleichgewichte zwischen Olivin (ol), Plagioklas (pl) und Klinopyroxen (cp) bei 1 atm beschreiben und die die Berechnung der Liquidus- Temperaturen für jedes dieser Minerale auf Basis der Schmelzzusammensetzung erlauben. Eine notwendige Voraussetzung zur Anwendung dieser Technik für die Abschätzung des H2O-Gehaltes ist die Sättigung der Gläser bezüglich of undpl bzw. für die Abschätzung des Kristallisationsdruckes bezüglichol undcp. Mineralogische Kriterien können zur Abschätzung der Sättigung herangezogen werden.
Die folgende Logik wird angewendet:
Die berechneten Liquidus-Temperaturen vonol, pl undcp müssen gleich sein wie für ein Glas, das abgeschreckte Schmelze repräsentiert aus dem diese Minerale bei 1 atm kristallisiert sind. Der Einfluß des Druckes resultiert hauptsächlich in einer Erhöhung dercp Kristallisations-Temperatur, im Vergleich zu der vonol undpl, wodurchcp gezwungen wird früher am Liquidus zu erscheinen. Für Kristallisation unter höherem Druck muß, als Folge der stärkeren Abhängigkeit dercp Kristallisationstemperatur vom Druck, die für 1 atm berechnete Temperatur voncp daher niedriger sein als die fürol. Die Differenz zwischen den bei 1 atm berechneten Liquidus-Temperaturen fürol undcp müssen daher den Kristallisationsdruck widerspiegeln. Es ist bekannt, daß H2O die Kristallisationstemperatur vonpl im Vergleich zuol undcp herabsetzt. Fürol-cp gesättigte H2O-führende Schmelzen muß daher die bei 1 atm und unter wasserfreien Bedingungen berechnete Temperatur vonpl höher sein als die fürol. Die Differenz zwischen diesen berechneten Temperaturen fürpl undol ergibt daher den H2O-Gehalt der Schmelze. p ]Da die Ergebnisse dieser Methode ausschließlich von der Qualität der Hauptelementanalysen der Gläser abhängen, weisen wir darauf hin, daß die Verwendung einer einzigen Glasanalyse zu falschen Ergebnissen führen kann. Die Verwendung von Mittelwerten, berechnet aus einer statistisch gesicherten Anzahl von Glasanalysen, wird daher bevorzugt. Wir weisen außerdem auf die Gefahr der Extrapolation dieser Technik auf Zusammensetzungen außerhalb des MORB—BABB Bereiches hin.
Similar content being viewed by others
References
Aggrey KE, Muenow DW, Batiza R (1988a) Volatile abundances in basaltic glasses from seamounts flanking the East Pacific Rise at 21 N and 12–14 N. Geochim Cosmochim Acta 52: 2115–2119
Aggrey KE, Muenow DW, Sinton JM (1988b) Volatile abundances in submarine glasses from the Northern Fiji and Lau back-arc basins. Geochim Cosmochim Acta 52: 2501–2506
Ariskin AA, Barmina GS (1990) Equilibria thermometry between plagioclases and basalt and andesite magmas. Geochem Int 27(10): 129–134
Ariskin AA, Barmina GS, Frenkel MYa (1987) Simulating low-pressure tholeiite-magma crystallization at a fixed oxygen fugacity. Geochem Int 24(5): 92–100
Baker DR, Eggler DH (1987) Compositions of anhydrous and hydrous melts coexisting with plagioclase, augite, and olivine or low-Ca pyroxene from I atm to 8 kbar: application to the Aleutian volcanic center of Atka. Am Mineral 72: 12–28
Bender JF, Hodges FN, Bence AE (1978) Petrogenesis of basalts from the project FAMOUS area: experimental study from 0 to 15 kbars. Earth Planet Sci Lett 41: 277–302
Byers CD, Muenow DW Garcia MO (1983) Volatiles in basalts and andesites from the Galapagos spreading center, 85 to 86 W. Geochim Cosmochim Acta 47: 1551–1558
Byers CD, Christie DM, Muenow DW, Sinton JM (1984) Volatile contents and ferric-ferros ratios of basalt, ferrobasalt, andesite and rhyodacite glasses from the Galapagos 95.5 W propagating rift. Geochim Cosmochim Acta 48: 2239–2245
Byers CD, Garcia MO, Muenow DW (1986) Volatiles in basaltic glasses from the East Pacific Rise at 21 N: implications for MORB sources and submarine lava flow morphology. Earth Planet Sci Lett 79: 9–20
Christie DM, Carmichael ISE, Langmuir CH (1986) Oxidation states of mid-ocean ridge basalt glasses. Earth Planet Sci Lett 79: 397–411
Danyushevsky LV, Sobolev AV, Kononkova NN (1992) Methods of studying magma inclusions in minerals during investigations on water-bearing primitive mantle melts (Tonga trench boninites). Geochem Int 29: 48–62
Danyushevsky LV, Falloon TJ, Sobolev AV, Crawford AJ, Carroll M, Price RC (1993) The H2O content of basalt glasses from Southwest Pacific back-arc basins. Earth Planet Sci Lett 117: 347–362
Delaney JR, Muenow DW Graham DG (1978) Abundance and distibution of water, carbon and sulfur in the glassy rims of submarine pillw basalts. Geochim Cosmochim Acta 42: 581–594
Duncan RA, Green DH (1987) The genesis of refractory melts in the formation of oceanic crust. Contrib Mineral Petrol 96: 326–342
Falloon TJ, Green DH (1987) Anhydrous partial melting of MORB pyrolite and other peridotite compositions at 10 kb: implications to the origin of primitive MORB glasses. Mineral Petrol 37: 181–219
Falloon TJ, Green DH (1988) Anhydrous partial melting of peridotite from 8 to 35 kb and the petrogenesis of MORB. J Petrol, Special Lithosphere Issue: 379–414
Falloon TJ, Green DH, Hatton, C J, Harris KL (1988) Anhydrous partial melting of a fertile and depleted peridotite from 2 to 30 kb and application to basalt petrogenesis. J Petrol 29: 1257–1282
Fisk MR, Bence AE (1980) Experimental crystallization of chrome spinel in FAMOUS basalt 527-1-1. Earth Planet Sci Lett 48: 111–123
Ford CE, Russell DG, Groven JA, Fisk MR (1983) Distribution coefficients of Mg2+, Fe2+, Ca2+ and Mn2+ between olivine and melt. J Petrol 24: 256–65
Fram MS, Longhi J (1992) Phase equilibria of dikes associated with Proterozoic anorthosite complexes. Am Mineral 77: 605–616
Fujii T, Scarfe CM (1985) Composition of liquids coexisting with spinel lherzolite at 10 kb and the genesis of MORB. Contrib Mineral Petrol 90: 18–28
Gaetani GA, Grove TL, Bryan WB (1994) Experimental phase relations of basaltic andesite from hole 839B under hydrous and anhydrous conditions. In:Hawkins JW, Parson L, Allan J, et al (eds) Proc ODP Sci Results 135. College Station, TX (Ocean Drilling Program), pp 557–565
Grove TL, Bryan WB (1983) Fractionation of pyroxene-phyric MOR at low pressure: an experimental study. Contrib Mineral Petrol 84: 293–309
Grove TL, Juster TC (1989) Experimental investigation of low-Ca pyroxene stability and olivine-pyroxene-liquid equilibria at 1-atm in natural basaltic liquids. Contrib Mineral Petrol 103: 287–305
Grove TL, Gerlach DC, Sando TW (1982) Origin of calc-alkaline series lavas at Medicine Lake volcano by fractionation, assimilation and mixing. Contrib Mineral Petrol 80: 160–182
Grove TL, Kinzler RJ, Bryan WB (1992) Fractionation of mid-ocean ridge basalt (MORB). In: Mantle flow and melt generation at mid-ocean ridges. AGU Geophysical Monograph 71, pp 281–310
Housh TB, Luhr JF (1991) Plagioclase-melt equilibria in hydrous system. Am Mineral 76: 477–492
Juster TC, Grove TL, Perfit MR (1989) Experimental constraints on the generation of FeTi basalts, andesites, and rhyodacites at the Galapagos spreading center, 85° W and 95° W. J Geophys Res 94B: 9251–9274
Langmuir CH, Klein EM, Plank T (1992) Petrological systematics of Mid-Ocean Ridge Basalts: constraints on melt generation beneath ocean ridges. In: Mantle flow and melt generation at mid-ocean ridges. AGU Geophysical Monograph 71, pp 183–280
Michael PJ, Chase RL (1987) The influence of primary magma composition, H2O and pressure on mid-ocean ridge basalt differentiation. Contrib Mineral Petrol 96: 245–263
Michael PJ, Schilling J-G (1989) Chlorine in mid-ocean ridge magmas: evidence for assimilation of sea-water-influenced components. Geochim Cosmochim Acta 53: 3131–3143
Muenow DW, Norman LKW, Garcia M0, Saunders AD (1980) Volatiles in submarine volcanic rocks from the spreading axis of the East Scotia Sea back-arc basin. Earth Planet Sci Lett 47: 272–278
Muenow DW Perfit MR, Aggrey KE (1991) Abundances of volatiles and genetic relationships among submarine basalts from the Woodlark Basin, southwest Pacific. Geochim Cosmochim Acta 55: 2231–2239
Nielsen RL, Dungan MA (1983) Low pressure mineral-melt equilibria in natural anhydrous mafic systems. Contrib Mineral Petrol 84: 310–326
Niu Y, Batiza R (1993) Chemical variation trends at fast and slow spreading Mid-Ocean Ridges. J Geophys Res 98B: 7887–7902
Poreda R (1985) Helium-3 and deuterium in back-arc basalts: Lau Basin and the Mariana Trough. Earth Planet Sci Lett 73: 244–254
Roeder PL, Emslie RF (1970) Olivine-liquid equilibrium. Contrib Mineral Petrol 29: 275–89
Sinton JM, Fryer P (1987) Mariana Trough lavas from 18°N: implications for the origin of back-arc basalts. J Geophys Res 92: 12782–12802
Sisson TW, Grove TL (1993) Experimental investigations of the role of H2O in calc-alkaline differentiation and subduction zone magmatism. Contrib Mineral Petrol 113: 143–166
Stolper E (1980) A phase diagram for mid-ocean ridge basalts: preliminary results and implications for petrogenesis. Contrib Mineral Petrol 74: 13–27
Takahashi E, Kushiro I (1983) Melting of a dry peridotite at high pressure and basalt magma genesis. Am Mineral 68: 859–79
Tormey DR, Grove TL, Bryan WB (1987) Experimental petrology of normal MORB near the Kane Fracture Zone - 22°–25°N, mid-Atlantic ridge. Contrib Mineral Petrol 96: 121–139
Volpe AM, MacDougall JD, Hawkins JW (1987) Mariana Trough basalts (MTB): trace element and Sr/Nd isotopic evidence for mixing between MORB-like and arc-like melts. Earth Planet Sci Lett 82: 241–254
Walker DA, Shibata T, Delong SE (1979) Abyssal tholeiites from the Oceanographer Fracture Zone. 11. Phase equilibria and mixing. Contrib Mineral Petrol 70: 111–125
Weaver JS, Langmuir CH (1990) Calculation of phase equilibrium in mineral-melt systems. Comp Geosci 16:1–19
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Danyushevskyl, L.V., Sobolevz, A.V. & Dmitrievz, L.V. Estimation of the pressure of crystallization and H2O content of MORB and BABB glasses: calibration of an empirical technique. Mineralogy and Petrology 57, 185–204 (1996). https://doi.org/10.1007/BF01162358
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01162358