Abstract
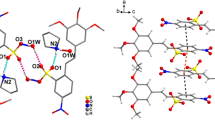
The crystal and molecular structure of 1,1′-bis-(2-naphthol) sulfone monopyridine (3) determined by single-crystal X-ray diffraction is reported. C25H19NO4S crystallizes in the monoclinic space groupC2/c (No. 15) witha=17.095(4),b=14.574(2),c=18.731(2) Å,β=116.29(2)°,Z=8,D x =1.364 Mg m−3,D m =1.353(8) Mg m−3. The phase problem was solved by direct methods and the final reliability factor obtained from full-matrix least-squares refinement of 2306 independent reflections is 0.067. The unit cell contains eight molecules with intramolecular hydrogen bonding and the molecules in the crystal lattice are held together by van der Waals forces. Conoscopic, infrared, and1H and C13 NMR analyses have been performed. Details of the structural content and selected bond lengths and angles are discussed.
Similar content being viewed by others
References
Bidics (1969–1981) Institute for Material Research, Hamilton, Canada.
Courriere, P., LaCroix, R., and Poupelin, J. (1979)Eur. J. Med. Chim Ther. 14 (1), 17–22.
Enraf-Nonius (1983)Structure determination package. (Enraf-Nonius, Delft, The Netherlands).
International Tables for X-ray Crystallography (1974) Vol. IV (Kynoch Press, Birmingham, England), pp. 99, 149.
Jones, P. Y. (1982) Thesis, Texas Southern University.
Orpen, A. G., Brammer, L., Allen, F. H., Kennard, O., Watson, D. G., and Taylor, R. (1989)J. Chem. Soc. Dalton Trans., S1.
SHELXTL-PC (1989)Seimens analytical x-ray instruments. (Madison, WI).
Shen, T. Y. (1964)International symposium on Nonsterodial inflammatory drugs. (Milan, Italy).
Yamaguchi, A., Yamaguchi, K., Murakami, H., and Kobayashi, T. (1981) U.S. Patent 4,287,366, Mitsui Toatsi Chemical, Inc.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mullica, D.F., Jones, J.W., Jones, D.L. et al. Synthesis, spectroscopic studies, and crystal structure of the first monopyridinium salt of 1,1′-bis-(2-naphthol) sulfone, C25H19NO4S. Journal of Crystallographic and Spectroscopic Research 23, 37–40 (1993). https://doi.org/10.1007/BF01161285
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01161285