Conclusions
-
1.
The ethyl ester of glycine in the copper complex of the Schiff base with salicylaldehyde is significantly more reactive than glycine in the copper complex of N-salicylideneglycinate.
-
2.
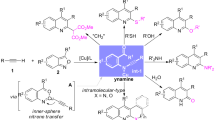
Aldehydes are added under mild conditions to bis[(N-carbethoxymethyl)salicylideneamino]copper, resulting in the formation of β-hydroxyamino acids.
-
3.
The reaction of bis[(N-carbethoxymethyl)salicylideneamino]copper with olefins with an activated double bond proceeds according to a scheme involving a Michael condensation followed by intramolecular cyclization, which results in the synthesis of 4-substituted 5-(2′-hydroxyphenyl)proline systems.
Similar content being viewed by others
Literature cited
Yu. N. Belokon', V. M. Belikov, N. I. Kuznetsova, and M. M. Dolgaya, Izv. Akad. Nauk SSSR, Ser. Khim., 1338 (1972).
Yu. N. Belokon', N. I. Kuznetsova, V. M. Belikov, arid R. L. Murtazin, Izv. Akad. Nauk SSSR, Ser. Khim., 2288 (1972).
Yu. N. Belokon', N. I. Kuznetsova, R. L. Murtazin, M. M. Dolgaya, Ts. B. Korchemnaya, and V. M. Belikov, Izv. Akad. Nauk SSSR, Ser. Khim., 2772 (1972).
Yu. N. Belokon', M. M. Dolgaya, N. I. Kuznetsova, S. V. Vitt, and V. M. Belikov, Izv. Akad. Nauk SSSR, Ser. Khim., 157 (1973).
I. P. Greenstein and M. Winitz, Chemistry of the Amino Acids, Vol. 3, Wiley, New York-London (1963), p. 2323.
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 4, pp. 890–894, April, 1977.