Abstract
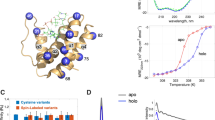
Fatty acid-binding protein (FABP) from bovine heart, a 15 kDa cytoplasmic protein has been investigated by multidimensional homonuclear and heteronuclear NMR-spectroscopy. Perdeuterated palmitic acid has been used as fatty acid ligand. The tertiary structure has been determined from distance geometry calculations with the variable target functions algorithm (DIANA) [1] utilizing 1027 interproton distance constraints, which were obtained from1H-homo-nuclear NOESY spectra. Overlapping NOE crosspeaks were assigned by heteronuclear multidimensional NMR-experiments with a15N-labelled sample. The tertiary structure resembles a β-barrel (β-clam) consisting of ten anti-parallel β-strands and a short helix-turn-helix motif. The β-strands are arranged in two nearly orthogonal β-sheets composed of 5 strands each. The solution structure is compared with the x-ray cyrstal structure of bovine heart [4] and rat intestinal FABPs.
Similar content being viewed by others
Abbreviations
- DOF-COSY:
-
Double Quantum Filtered Correlated Spectroscopy
- TOCSY:
-
Total Correlated Spectroscopy
- NOE:
-
Nuclear Overhauser Enhancement
- NOESY:
-
Nuclear Overhauser Enhancement and Exchange Spectroscopy
- HMQC:
-
Heteronuclear Multiple Quantum Coherence
- FABP:
-
Fatty Acid-Binding Protein
- FABPc :
-
Cellular Fatty Acid-Binding Protein
- H-FABPc :
-
Cellular Heart Fatty Acid-Binding Protein
- I-FABPc :
-
Cellular Intestinal Fatty Acid-Binding Protein
References
Güntert P, Braun W, Wüthrich K: Efficient computation of three-dimensional protein structures in solution from NMR data using the program DIANA and the supporting programs CALIBA, HABAS and GLOMSA. J Mol Biol 217: 517–530, 1991
Bass NM: The cellular fatty acid-binding proteins: aspects of structure, regulation and function. Internat Rev Cytol 111: 143–185, 1988
Veerkamp JH, Peeters RA, Maatman RGHJ: Structural and functional features of different types of cytoplasmic fatty acid-binding proteins. BBA 1081: 1–24, 1991
Müller-Fahrnow A, Egner U, Jones TA, Rüdel H, Spener F, Sänger W: Three-dimensional structure of fatty acid-binding protein from bovine heart. Eur J Biochem 199: 271–276, 1991
Sacchettini JC, Gordon JI, Banaszak LJ: Crystal structure of rat intestinal fatty acid-binding protein. J Mol Biol 208: 327–339, 1989
Scapin G, Gordon JI, Sacchettini JC: Refinement of the structure of recombinant rat intestinal fatty acid-binding apoprotein at 1.2 Å resolution. J Biol Chem 267: 4253–4269, 1992
Scapin G, Spandon P, Mammi M, Zanotti G, Monaco HL: Crystal structure of chicken liver basic fatty acid-binding protein at 2.7 Å resolution. Mol Cell Biochem 98: 95–99, 1990
Jones TA, Bergfors T, Sedzik J, Unge T: The three-dimensional structure of P2 myelin protein. EMBO J 7: 1597–1604, 1988
Unterberg C, Börchers T, Højrup P, Roepstorff P, Knudsen J, Spener F: Cardiac fatty acid-binding proteins. J Biol Chem 265: 16255–16261, 1990
Billich S, Wissel T, Kratzin H, Hahn U, Hagenhoff B, Lezius A, Spener F: Cloning of a full-length complementary DNA for fatty acid-binding protein from bovine heart. Eur J Biochem 175: 549–556, 1988
Schulenberg-Schell H, Schäfer P, Keuper HJK, Stanislawski B, Hoffmann E, Rüterjans H, Spener F: Interactions of fatty cids with neutral fatty acid-binding protein from bovine liver. Eur J Biochem 170: 565–574, 1988
Cistola DP, Sacchettini JC, Gordon JI:13C NMR studies of fatty acid-protein interactions: comparison of homologous fatty acid-binding proteins produced in the intestinal epithelium. Mol Cell Biochem 98: 101–110, 1990
Lücke C, Lassen D, Kreienkamp H-J, Spener F, Rüterjans H: Sequence-specific1H NMR assignment and determination of the secondary structure of bovine heart fatty acid-binding protein. Eur J Biochem 210: 901–910, 1992
Unterberg C, Heidl G, von Bassewitz D-B, Spener F: Isolation and characterization of the fatty acid-binding protein from human heart. J Lipid Res 27: 1287–1293, 1986
Glatz JFC, Veerkamp JH: A radiochemical procedure for the assay of fatty acid-binding by proteins. Anal Biochem 132: 89–95, 1983
Shon K, Opella SJ: Detection of1H homonuclear NOE between amide sites in proteins with1H/15N heteronuclear correlation spectroscopy. J Magn Reson 82: 193–197, 1989
Fesik SW, Zuiderweg ERP: Heteronuclear three-dimensional NMR spectroscopy. A strategy for the simplification of homonuclear two-dimensional NMR spectra. J Magn Reson 78: 588–593, 1988
Marion D, Kay LE, Sparks SW, Torchia DA, Bax A: Three-dimensional heteronuclear NMR of15N-labeled proteins. J Am Chem Soc 111: 1515–1517, 1989
Kay LE, Marion D, Bax A: Practical aspects of 3D heteronuclear NMR of proteins. J Magn Reson 84: 72–84, 1989
Zuiderweg ERP: A proton-detected heteronuclear chemical-shift correlation experiment with improved resolution and sensitivity. J Magn Reson 86: 346–357, 1990
Güntert P, Wüthrich K: Improved efficiency of protein structure calculations from NMR data using the program DIANA with redundant dihedral angle constraints. J Biomol NMR 1: 447–456, 1991
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lassen, D., Lücke, C., Kromminga, A. et al. Solution structure of bovine heart fatty acid-binding protein (H-FABPc). Mol Cell Biochem 123, 15–22 (1993). https://doi.org/10.1007/BF01076470
Issue Date:
DOI: https://doi.org/10.1007/BF01076470