Summary
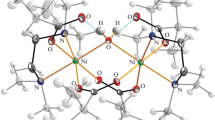
The formation equilibria of nickel(II) violurato-complexes in water and dimethylsulphoxide-water (80∶20) as well as deprotonation of the coordinated ligands and related stereochemical changes are reported. The stability constants of the Ni2+-H2V− complexes logβ1=5.06, Iogβ2=9.38, logβ3=12.98 as well as the acidity constants of the [Ni(H2V)3]− complex, logβj1= 8.37, logβj2=15.76, logβj3=22.37 are determined at 25 °C and 0.1M NaClO4. A new violurato-complex of Ni2+, Na4[Ni(HV)3]5H2O, is isolated and characterized.
Similar content being viewed by others
References
E. García-España, J. Moratal and J. Faus,J. Coord. Chem., 12, 41 (1982) and refs. therein.
J. Moratal, J. Faus and M. Juive,Rev. Chim. Min., 19, 72 (1982).
J. Moratal, A. Prades, M. Juive and J. Faus,Thermochim. Acta, 89, 343 (1985).
J. Moratal and J. Faus,Inorg. Chim. Acta, 25, L1 (1977).
C. Bremard, G. Nowogrocki and S. Sueur,J. Chem. Soc., Dalton Trans., 1856 (1981).
J. Moratal and J. Faus,Rev. Chim. Min., 16, 203 (1979).
J. Faus, J. Moratal and M. Juive,J. Inorg. Nucl. Chem., 42, 1786 (1980).
P. Gans, A. Sabatini and A. Vacca,J. Chem. Soc., Dalton Trans., 1195 (1985).
D. Perrin and B. Dempsey,Buffers for pH and Metal Ion Control, Chapman and Hall, London, 1974, p. 147.
E. Asmus,Z.Anal. Chem., 178, 104 (1960).
M. Ceresole,Chem. Ber., 16, 1133 (1888).
F. Lloret, J. Moratal and J. Faus,J. Chem. Soc., Dalton Trans., 1743 (1983).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
García-España, E., Moratal, J.M. & Faus, J. Violurato complexes of nickel(II). Formation equilibria. Deprotonation equilibria of the coordinated ligands and related stereochemical changes. Transition Met Chem 11, 1–5 (1986). https://doi.org/10.1007/BF01064490
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01064490