Summary
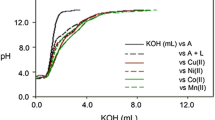
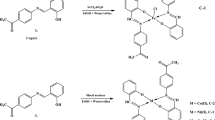
CoII, NiII, CuII, CdII and HgII complexes of 7-formyl-8-hydroxyquinoline (HFHQ) have been prepared, and characterized by elemental analysis, molar conductivities, electronic and i.r. spectra, and magnetic measurements. It was found that the ligand acts as a neutral monodentate or a monobasic bidentate donor. The CoII, NiII and CuII complexes possess, respectively, tetrahedral, octahedral and square-planar structures based on their magnetic and electronic spectral data. The electrical conductivities of HFHQ and its metal complexes were measured at different temperatures and their activation energies were calculated. The values obtained for the CoII, NiII, CdII and HgII complexes agree fairly well with those reported for semiconductors. The ligand was tested as a corrosion inhibitor for aluminium; the limiting concentration of HFHQ to give maximum efficiency (99.2%) is 10−3 mole dm−3 at 22°C. The metal-ligand ratios and apparent formation constants for the species generated in ethanol solution were determined spectrophotometrically.
Similar content being viewed by others
References
E. Proffit and G. Buchmann,Arznemittel Forsch,10, 181 (1960).
Hoffman La Roche, Swiss patent, 416648 (1967).
J. A. Vaichulis, U. S. patent 3272352 (1966).
H. Dietrich, Swiss patent, 454874 (1968).
L. H. Schmidt,Ann. Rev. Microbiol.,427, 23 (1969).
S. Aktiesel and F. Kabet, Dan. patent, 108626 (1968).
D. R. Williams,Chem. Rev.,72, 203 (1972).
A. M. Hammam, A. Awad and H. Saber,J. Ind. Chem. Soc.,58(2), 184 (1981).
M. R. Mahmoud, A. Awad, A. M. Hamman and H. Saber,Ind. J. Chem. 19A, 1131 (1980).
R. Feigl,Spot Test in Organic Analysis, Elsevier, Amsterdam, 1966.
W. J. Geary,Coord. Chem. Rev.,7, 81 (1971).
J. I. Bullock and H. A. Tajmir-Riahi,J. Chem. Soc., Dalton Trans., 34 (1978).
J. R. Ferraro,Low Frequency Vibrations of Inorganic and Coordination Compounds, Plenum Press, New York, 1971.
N. Nakamoto,Infrared Spectra of Inorganic and Coordination Compounds, Wiley, New York, 1970.
A. N. Speca, N. M. Karayanis and L. H. Pytlewiski,Inorg. Chem. Acta,9, 87 (1974).
A. B. P. Lever,Inorganic Electronic Spectroscopy, Elsevier, Amsterdam, 1968, p. 341.
M. E. Khalifa, A. A. El-Asmy, K. M. Ibrahim and M. M. Mostafa,Synth. React. Inorg. Met-Org. Chem.,16, 1305 (1986).
M. M. Mostafa, A. S. El-Asmy and G. M. Ibrahim,Transition Met. Chem.,8, 54 (1983).
M. Pulaniandavar and C. Natarajan,Aust. J. Chem.,33, 737 (1980).
B. Bosnich,J. Am. Chem. Soc.,90, 627 (1968).
W. C. Vosburgh and G. R. Cooper,J. Am. Chem. Soc.,63, 937 (1941).
A. A. El-Asmy, Y. M. Shaibi, A. S. Babaqi, M. Mounir and S. A. Ashour,Trans. Met. Chem.,13(5), 332 (1988).
P. D. Ankrum,Semiconductor Electronics, Prentice Hall Inco., India, 1981.
R. N. Sen and S. K. Ray,J. Chem. Soc., (A), 1658 (1932).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
El-Asmy, A.A., El-Sonbati, A.Z., Ba-Issa, A.A. et al. Synthesis and properties of 7-formyl-8-hydroxyquinoline and its transition metal complexes. Transition Met Chem 15, 222–225 (1990). https://doi.org/10.1007/BF01038379
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01038379