Summary
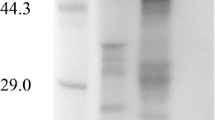
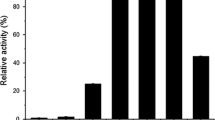
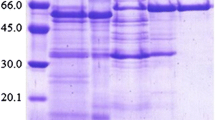
Two extracellular α-glucosidases (EC 3.2.1.20, α-D-glucoside glucohydrolase) of the alkalophilic bacterium,Bacillus sp. NCIB 11203, were separated, purified and partially characterised. Resolution of the system into two separate enzymes was achieved by fractionation with (NH4)2SO4 and chromatography on DEAE-Biogel A. The first of these activities, an α-glucosidase, hydrolysed p-nitrophenyl-α-D-glucopyranoside preferentially and had minor activity on isomaltose and isomaltotriose. The second enzyme was a maltase and displayed highest activity on maltose and maltotriose and some activity on p-nitrophenyl-α-D-glucopyranoside.
Similar content being viewed by others
References
Boyer, E.W. and Ingle, M.B. (1972). J. Bacteriol.110:992–1000.
Fogarty, W.M. (1983). Microbial amylases.In Microbial enzymes and biotechnology. Chap. 1.Edited by W. M. Fogarty. Applied Science Publishers, London. pp. 1–92.
Fogarty, W.M., Kelly, C.T. and Kadam, S.K. (1985). Can. J. Microbiol.31: 670–674.
Huggett, A.S.C. and Nixon, D.A. (1955). Biochem. J.66: 12–19.
Kelly, C.T. and Fogarty, W.M. (1983). Process Biochem.18: 6–12.
Kelly, C.T., Giblin, M. and Fogarty, W.M. (1986). Can. J. Microbiol.32: 342–347.
Kelly, C.T., Moriarty, M. and Fogarty, W.M. (1983a). Biotechnol. Lett.5: 289–294.
Kelly, C.T., O'Reilly, F. and Fogarty, W.M. (1983b). FEMS Microbiol. Lett.20: 55–59.
Lowry, D.H., Rosebrough, N.J., Farr, A.L. and Randall, R.J. (1951). J. Biol. Chem.193: 265–275.
Thirunavukkarasu, M. and Priest, F.G. (1984). J. Gen. Microbiol.130: 3135–3141.
Urlaub, H. and Wöber, G. (1978). Biochim. Biophys. Acta,522: 161–173.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kelly, C.T., Brennan, P.A. & Fogarty, W.M. Resolution of the extracellular α-glucosidase system ofBacillus sp. NCIB 11203. Biotechnol Lett 9, 125–130 (1987). https://doi.org/10.1007/BF01032751
Issue Date:
DOI: https://doi.org/10.1007/BF01032751