Abstract
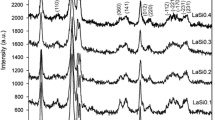
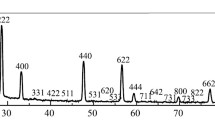
The behaviour of three different samples of La2O3 on exposure to atmoshperic CO2 and H2O and the influence of the origin is discussed. The thermal evolution of the samples, stabilized in air for months or even years, has been studied by TG, TPD, IR spectroscopy and X-ray diffraction. BET surface areas of the samples were determined from the corresponding nitrogen adsorption isotherms at 77 K. In all three samples, hydration and carbonation occur in bulk. In accordance with our results, lanthana samples stabilized in air would consist of lanthanum hydroxide, La(OH)3, partially carbonated, La2(OH)6−3x (CO3) x ,(x⪝1). When hexagonal phases of La2O3, obtained by calcining, at 1130 K, the samples stabilized in air were re-exposed, hydration and carbonation levels similar to those observed in the stabilized samples were reached after less than 24 h. Some lanthana samples were soaked in water, at 298 K, and then dried at 380 K. In this way, the evolution of the oxide when treated under similar conditions to those used in both impregnation and ion exchange techniques for preparation of supported metal phases, could be investigated.
Similar content being viewed by others
References
M. W. Shaffer andR. Roy,J. Amer. Ceram. Soc. 42 (1959) 503.
J. M. Haschke andL. Eyring,Inorg. Chem. 10 (1971) 2267.
B. H. T. Chai andS. Mroczkowski,J. Cryst. Growth 44 (1978) 84.
M. N. Viswanatiah andJ. A. K. Tareen,Mater. Res. Bull. 15 (1980) 855.
H. T. Fullam andF. P. Roberts, US Atomic Energy Commission, Report AT (45-1)-1830 (1970) p. 1.
S. Bernal, F. J. Botana, R. García andJ. M. Rodríguez-Izquierdo,Termochim. Acta 66 (1983) 139.
S. Bernal, R. García, J. M. Rodriguez-Izquierdo andJ. M. Trillo,J. Less Common Met. 94 (1983) 145.
S. Bernal, R. García, J. M. Pintado andJ. M. Rodríguez-Izquierdo. Eighth National Meeting on Adsorption. Málaga, Spain, (1983) p.117.
R. Alvero, Doctoral thesis, University of Seville (1983).
J. R. Anderson, “Structure of Metallic Catalysts” (Academic Press, London, 1975).
J. P. Brunelle,Pure Appl. Chem. 50 (1978) 1211.
R. Alvero, J. A. Odriozola, J. M. Trillo andS. Bernal.J. Chem. Soc. Dalton (1984) 87.
R. J. Cvetanovic andY. Amenomiya,Adv. Catal. 17 (1967) 103.
M. P. Rosynek andD. T. Magnuson,J. Catal. 46 (1977) 402.
N. V. Zubova, V. N. Makarov, V. D. Nikolskii, P. N. Petrov, E. G. Teterin andN. T. Chebotarev,Russ. J. Inorg. Chem. 13 (1968) 7.
S. Bernal, R. García, J. M. López andJ. M. Rodr∥uez-Izquierdo,Collect. Czech. Chem. Commun. 48 (1983) 2205.
P. Caro andM. Lamaitre Blaise,Compt. Rend. Acad. Sci. Paris, ser. C 269 (1969) 687.
P. Caro, J. C. Achard andO. Pous,Colloque International du CNRS sur les elements des terres rares 1 (1970) 285.
P. E. Caro, J. O. Sawyer andL. Eyring,Spectrochim. Acta 28 (1972) 1167.
R. P. Turcotte, J. M. Haschke, M. S. Jenkins andL. Eyring,J. Solid State Chem. 2 (1970) 593.
I. S. Shplygin, V. P. Komarov andV. B. Lazarev,J. Therm. Anal. 15 (1979) 215.
V. V. S. Rao, R. V. G. Rao andA. B. Biswas,J. Inorg. Nucl. Chem. 27 (1965) 2525.
R. P. Turcotte, J. O. Sawyer andL. Eyring,Inorg. Chem. 8 (1969) 238.
E. Ramaroson, R. Kieffer andA. Kiennemann,J. Chem. Soc. Chem. Commun. (1982) 645.
P. R. Watson andG. A. Somorjai,J. Catal. 74 (1982) 282.
R. F. Hicks, Y. Q. Jie andA. T. Bell, Eighth North American Meeting of the Catalysis Society, Paper B-21, Philadelphia (1983).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bernal, S., Díaz, J.A., García, R. et al. Study of some aspects of the reactivity of La2O3 with CO2 and H2O. J Mater Sci 20, 537–541 (1985). https://doi.org/10.1007/BF01026524
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01026524