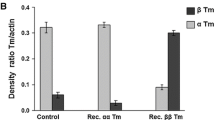
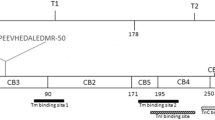
Abstract
The regulatory roles of the components of the troponin-tropomyosin complex in the presence and absence of Mg2+ on the acto-S1 ATPase have been examined. The effect of free Mg2+ on the inhibition of the acto-S1 ATPase by rabbit skeletal troponin (Tn) was studied at S1 to actin ratios ranging from 0.17:1 to 2.5:1. These studies were performed using two Mg2+ concentrations: 2.5 mM Mg2+-2.5 mM ATP, conditions considered to have low free Mg2+; and 5.0 mM Mg2+-2.5 mM ATP, conditions providing a high free Mg2+ concentration of ∼2.5 mM. In the presence of high free Mg2+ (2.5 mM ATP-5.0 mM MgCl2) the Tn inhibition of acto-S1-TM ATPase increased by approximately 40–50% over a range of S1 to actin ratios of 0.17:1 to 2.5:1. The effect of free Mg2+ on increasing quantities of Tn in the absence or presence of tropomyosin was studied independently at two S1 to actin ratios (1:1 and 2:1). In the absence of TM, at 5 mM Mg2+ there is an additional 38% (1:1 S1 to actin) or 37% (2:1) decrease in the ATPase activity by Tn compared to 2.5 mM Mg2+. Similarly, in the presence of TM and Tn, Mg2+ exerts its effect at both S1 to actin ratios. Significantly, the inhibition by the IT complex in the presence of TM is unaffected by free Mg2+. Furthermore, ultracentrifugation binding studies using14C-iodoacetamide-labeled Tn and TM established that the Tn-TM regulatory complex was firmly bound to F-actin at both Mg2+ concentrations, indicating that faciliation of binding to F-actin by Mg2+ is not responsible for the increased inhibition. Hence, it is concluded from the data that Mg2+ binding and by analogy Ca2+ binding to the Ca2+-Mg2+ sites of TnC promotes muscle relaxation by inducing inhibition of the actomyosin ATPase, whereas Ca2+ binding to the Ca2+-specific sites promotes contraction by potentiating the ATPase. The inhibition of the acto-S1-TM ATPase by TnT has also been further examined. The data indicate that TnT exerts the same level of inhibition upon the ATPase as TnI or Tn. The inhibitory activity requires TM, and occurs to the same extent under conditions where TM alone would have either a potentiating (2:1 S1 to actin) or an inhibitory (1:1 S1 to actin) effect upon the ATPase. In the presence of TM the IT complex is a more effective inhibitor than either TnI, TnT, or Tn. The inhibitory activity of the IT complex is partially released by TnC in the absence of Ca2+. These observations, in conjunction with those by Chong, Asselbergs, and Hodges, which showed that the inhibition by TnT is partially released by TnC plus Ca2+, indicate that the role of TnT involves more than anchoring Tn to the thin filament.
Similar content being viewed by others
References
Bremel, R. D., Murray, J. M., and Weber, A. (1972).Cold Spring Harbor Symp. Quant. Biol. 37, 267.
Chong, P. C. S., and Hodges, R. S. (1982a).J. Biol. Chem. 257, 2549.
Chong, P. C. S., and Hodges, R. S. (1982b).J. Biol. Chem. 257, 9152.
Chong, P. C. S., Asselbergs, P. J., and Hodges, R. S. (1983).FEBS Lett. 153, 372.
Coté, G. P., and Smillie, L. B. (1981).J. Biol. Chem. 256, 11999.
Cox, J. A., Comte, M., and Stein, E. A. (1981).Biochem. J. 195, 205.
Dabrowska, R., Podlubnaya, Z., Nowak, E., and Drabikowski, W. (1976).J. Biochem. 80, 89.
Drabikowski, W., and Dabrowska, R. (1975). InProceedings of the IXth FEBS Meeting, Proteins of Contractile Systems (Biro, E. N. A., ed.), Akademiai Kaido, Budapest/North-Holland Amsterdam, Vol. 31, pp. 85–103.
Drabikowski, W., Barylko, B., Dabrowska, R., Nowak, E., and Szpacenko, A. (1974). InCalcium Binding Proteins (Drabikowski, W., Strzdecka-Golaszewska, H., and Carafoli, E., eds.), Elsevier, Amsterdam, pp. 69–107.
Eaton, B. L., Kominz, D. R., and Eisenberg, E. (1975).Biochemistry 14, 2718.
Eisenberg, E., and Kielley, W. W. (1974).J. Biol. Chem. 249, 4742.
Frieden, C., Lieberman, D., and Gilbert, H. R. (1980).J. Biol. Chem. 255, 8991.
Grabarek, Z., Drabikowski, W., Leavis, P. C., Rosenfeld, S. S., and Gergely, J. (1981).J. Biol. Chem. 256, 13121.
Greaser, M. L., and Gergely, J. (1971).J. Biol. Chem. 246, 4226.
Greaser, M. L., Yamaguchi, M., Brekke, C., Potter, J., and Gergeley, J. (1972).Cold Spring Harbor Symp. Quant. Biol. 37, 235.
Haselgrove, J. C. (1972).Cold Spring Harbor Symp. Quant. Biol. 37, 341.
Head, J. F., and Perry, S. V. (1974).Biochem. J. 137, 145.
Herzberg, O., and James, M. N. G. (1985).Nature 313, 653.
Hill, T. E., Eisenberg, E., and Greene, L. E. (1983).Proc. Natl. Acad. Sci. USA 80, 60.
Hitchcock, S. E. (1981).Mol. Biol. 147, 153.
Hitchcock, S. E., Zimmerman, C. J., and Smalley, C. (1981).J. Mol. Biol. 147, 125.
Hitchcock-DeGregori, S. E. (1982).J. Biol. Chem. 257, 7372.
Horwitz, J., Bullard, B., and Mercola, D. (1979).J. Biol. Chem. 254, 350.
Huxley, H. E. (1972).Cold Spring Harbor Symp. Quant. Biol. 37, 361.
Ingraham, R. H. (1984). Ph.D. Thesis, University of Iowa, Iowa City, Iowa.
Ingraham, R. H., and Swenson, C. A. (1983).Eur. J. Biochem. 132, 85.
Ingraham, R. H., and Swenson, C. A. (1984).J. Biol. Chem. 259, 9544.
Ishiwata, S. (1978).Biochim. Biophys. Acta 534, 350.
Jackson, P., Amphlett, G. W., and Perry, S. V. (1975).Biophys. J. 151, 85.
Johnson, J. D., and Potter, J. D. (1978).J. Biol. Chem. 253, 3775.
Johnson, J. D., Charlton, S. C., and Potter, J. D. (1979).J. Biol. Chem. 254, 3497.
Katayama, E. (1980). InMuscle Contraction: Its Regulatory Mechanisms (Ebashi, S., Maruyama, K., and Endo, M., eds.), Japan Scientific Society Press, Tokyo/Springer-Verlag, Berlin, pp. 251–258.
Kawasaki, Y., and van Eerd, J. (1972).Biochem. Biophys. Res. Commun. 49, 898.
Leavis, P. C., and Gergely, J. (1984).CRC Crit. Rev. Biochem. 16, 235.
Leavis, P. C., Rosenfeld, S. S., Gergely, J., Grabarek, Z., and Drabikowski, W. (1978).J. Biol. Chem. 253, 5452.
Lehrer, S. S., and Leavis, P. C. (1974).Biochem. Biophys. Res. Commun. 58, 159.
Lehrer, S. S., and Morris, P. (1982).J. Biol. Chem. 257, 8073.
Lohmeier, E. M. (1982). Ph.D. Thesis, University of Alberta, Edmonton, Alberta, Canada.
Mak, A. S., and Smillie, L. B. (1981).J. Mol. Biol. 149, 541.
Mak, A. S., Golosinska, K., and Smillie, L. B. (1983).J. Biol. Chem. 258, 14330.
Mani, R. S., McCubbin, W. D., and Kay, C. M. (1974).FEBS Lett. 38, 357.
Mani, R. S., McCubbin, W. D., and Kay, C. M. (1975).FEBS Lett. 52, 127.
Morris, E. P., and Lehrer, J. S. (1984).Biochemistry 23, 2214.
Nagy, B., and Gergely, J. (1979).J. Biol. Chem. 254, 12732.
Ohnishi, S., Maruyama, K., and Ebashi, S. (1975).J. Biochem. 78, 73.
Ohtsuki, I. (1979).J. Biochem. (Tokyo)86, 491.
Pato, M. B., Mak, A. S., and Smillie, L. B. (1981).J. Biol. Chem. 256, 593.
Pearlstone, J. R., and Smillie, L. B. (1978).Can. J. Biochem. 56, 521.
Pearlstone, J. R., and Smillie, L. B. (1981).FEBS Lett. 128, 119.
Pearlstone, J. R., and Smillie, L. B. (1982).J. Biol. Chem. 257, 10587.
Pearlstone, J. R., and Smillie, L. B. (1983).J. Biol. Chem. 258, 2534.
Potter, J. D., and Gergely, J. (1974).Biochemistry 13, 2697.
Potter, J. D., and Gergely, J. (1975).J. Biol. Chem. 250, 4628.
Potter, J. D., Seidel, J. C., Leavis, P., Lehrer, S. S., and Gergely, J. (1976).J. Biol. Chem. 251, 7551.
Potter, J. D., Robertson, S. P., Collins, J. H., and Johnson, J. D. (1980). InCalcium-Binding Proteins: Structure and Function (Siegel, F. L., Carafoli, A., Kretsinger, R. H., MacLennan, D. H., and Wasserman, R. H., eds.), Elsevier/North-Holland, Amsterdam, pp. 279–288.
Seamon, K. B., Hartshorne, D. J., and Bothner-By, A. A. (1977).Biochemistry 16, 4039.
Spudich, J. A., and Watts, S. (1971).J. Biol. Chem. 246, 4866.
Talbot, J. A., and Hodges, R. S. (1979).J. Biol. Chem. 254, 3720.
Talbot, J. A., and Hodges, R. S. (1981).J. Biol. Chem. 256, 2798.
Tsalkova, T. N., and Privalov, P. L. (1980).Biochim. Biophys. Acta 624, 196.
Van Eerd, J., and Kawasaki, Y. (1973).Biochemistry 12, 4972.
Weeds, A. G., and Taylor, R. S. (1975).Nature 257, 54.
Wegner, A. (1979).J. Mol. Biol. 131, 839.
Wegner, A., and Walsh, T. P. (1981).Biochemistry 20, 5633.
Wilkinson, J. M., Perry, S. V., Cole, H. A., and Trayer, I. P. (1972).Biophys. J. 127, 215.
Zot, H. G., and Potter, J. D. (1982).J. Biol. Chem. 257, 7678.
Zot, H. G., Guth, K., and Potter, J. D. (1985).Biophys. J. 47, 473a.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Van Eyk, J.E., Cachia, P.J., Ingraham, R.H. et al. Studies on the regulatory complex of rabbit skeletal muscle: Contributions of troponin subunits and tropomyosin in the presence and absence of Mg2+ to the acto-S1 ATPase activity. J Protein Chem 5, 335–354 (1986). https://doi.org/10.1007/BF01025962
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF01025962