Abstract
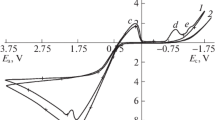
The electrochemical behaviour of synthetic silver sulphide (acanthite) electrodes in sulphuric acid solutions has been investigated using several techniques including cyclic voltammetry, anodic polarization and constant potential experiments. Under anodic polarization the dissolution has been attributed to the reaction Ag2S=2Ag++S+2e which occurs in two sequential, single electron transfer steps. A kinetic model for this stepwise anodic dissolution process at lower overpotential, where the current is a function of potential, is provided. At high dissolution rates (i.e. high currents) the slightly soluble silver sulphate salt is formed on the surface due to the saturation of the electrolyte near the Ag2S interface. This observation is supported by the influence of electrolyte composition on the cyclic voltammetry and the polarization curve. A paralinear film growth model has been found to describe the formation and growth of the silver sulphate product layer indicating an initial region of parabolic kinetics which gradually changes to linear kinetics as the rate of film dissolution approaches that of film formation.
Similar content being viewed by others
References
G. J. Snell and M. C. Sze,Eng. Mining. J. 178 (1977) 100.
F. E. Pawlek, in ‘Extractive Metallurgy of Copper’ (edited by J. C. Yannopoulos and J. C. Agarwal) AIME, New York (1976) p. 690.
J. D. Miller and H. Q. Portillo,Dev. Miner. Process. 2 (1981) 851.
G. W. Warren, B. Drouven and D. W. Price,Metall. Trans. B 15B (1984) 235.
R. L. Paul, M. J. Nicol, J. W. Diggle and A. P. Saunders,Electrochim. Acta 23 (1978) 625.
B. Dandapani and E. Ghali,Trans. Instn. Min. Metall. 91 (1982) 38.
R. A. Narasgoudar, J. W. Johnson, and T. J. O'Keefe,Hydrometallurgy 9 (1982) 37.
T. Biegler and D. A. Swift,J. Appl. Electrochem. 9 (1979) 545.
B. Dandapani and E. Ghali,J. Electrochem. Soc. 129 (1982) 271.
M. H. Lietzke and R. W. Stoughton,J. Phys. Chem. 65 (1961) 2247.
,J. Amer. Chem. Soc. 78 (1956) 3023.
J. Gulens and D. W. Shoesmith,J. Electrochem. Soc. 128 (1981) 811.
V. I. Birss and G. A. Wright,Electrochim. Acta 27 (1982) 1.
J. O'M. Bockris and G. A. Razumney, ‘Fundamental Aspects of Electrocrystallization’, Plenum Press, New York (1967) p. 36.
J. B. Hiskey,Inst. Min. Metall. 88C (1979) 145.
M. J. Nicol, R. L. Paul and J. W. Diggle,Electrochim. Acta 23 (1978) 635.
K. J. Vetter, ‘Electrochemical Kinetics’, Academic Press, New York (1967) pp. 149–53.
M. E. Wadsworth and T. K. Zhong, in ‘Hydrometallurgical Process Fundamentals’ (edited by R. G. Bautista), Plenum Press, New York (1984).
J. O'M. Bockris and A. K. N. Reddy, ‘Modern Electrochemistry’, Plenum Publishing Co., New York (1973) pp. 1291–5.
A. R. Despic and J. O'M. Bockris,J. Chem. Phys. 32 (1960) 389.
P. Kofstad, ‘High Temperature Oxidation of Metals’, John Wiley, New York (1966) pp. 1–19.
J. H. Ahn, PhD dissertation, Department of Metallurgical Engineering, University of Utah, Salt Lake City, Utah (1985).
E. W. Haycock,J. Electrochem. Soc. 106 (1959) 771.
G. W. Warren and M. E. Wadsworth,Metall. Trans. B 15B (1984) 289.
D. W. Price and G. W. Warren,Hydrometallurgy 15 (1986) 303.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Price, D.W., Warren, G.W. & Drouven, B. The electrochemical behaviour of silver sulphide in sulphuric acid solutions. J Appl Electrochem 16, 719–731 (1986). https://doi.org/10.1007/BF01006925
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF01006925