Conclusions
-
1.
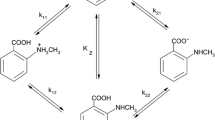
A tautomeric equilibrium is established between the neutral and zwitterion forms of the ortho-aminomethylphenols in aqueous solutions.
-
2.
Determination has been made of the tautomeric equilibrium constants and the acid-base equilibrium constants for the various forms of the ortho-aminomethylphenols.
-
3.
An equation for calculating the tautomeric equilibrium constant has been developed.
Similar content being viewed by others
Literature cited
A. M. Kuliev, M. A. Salimov, F. N. Mamedov, and N. Yu. Ibragimov, Dokl. Akad. Nauk SSSR,184, 1141 (1969).
V. P. Lezina, V. F. Bystrov, B. E. Zaitsev, N. A. Andronova, L. D. Smirnov, and K. M. Dyumaev, Teor. Eksp. Khim.,5, 247 (1969).
V. E. Bel'skii, L. A. Kudryavtseva, N. A. Shishkina, T. A. Zyablikova, A. V. Il'yasov, and B. E. Ivanov, Izv. Akad. Nauk SSSR, Ser. Khim., 331 (1977).
L. Sucha, Z. Urner, and M. Suchanek, Collect. Czech. Chem. Commun.,35, 3651 (1970).
D. H. McDaniel and H. C. Brown, J. Am. Chem. Soc.,77, 3756 (1955).
N. A. Shishkina, K. A. Derstuganova, L. A. Kudryavtseva, V. E. Bel'skii, and B. E. Ivanov, Izv. Akad. Nauk SSSR, Ser. Khim., 1259 (1976).
B. A. Korolev, M. A. Mal'tseva, A. I. Tarasov, and V. A. Vasnev, Zh. Obshch. Khim.,46, 864 (1974).
H. K. Hall, J. Am. Chem. Soc.,79, 5441 (1957).
J. J. Christensen, D. P. Wrathall, R. M. Jzatt, and D. O. Tolman, J. Phys. Chem., 3001 (1967).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 4, pp. 803–808, April, 1980.
Rights and permissions
About this article
Cite this article
Teitel'baum, A.B., Derstuganova, K.A., Shishkina, N.A. et al. Tautomerism in the ortho-aminomethylphenols. Russ Chem Bull 29, 558–562 (1980). https://doi.org/10.1007/BF00961603
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00961603