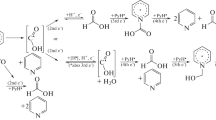
Conclusions
-
1.
The throughgoing electrochemical oxidation of esters of malonic acid in the presence of catalyst carriers — salts of hydriodic acid — in methanol and ethanol leads to esters of ethylenetetracarboxylic acid and the products of the addition to it of alcohols and dialkyl malonates, namely 1-alkoxyethane-1,1,2,2-tetracarboxylic and propane-1,1,2,2,3,3-hexacarboxylic esters.
-
2.
The results of the reaction depends to a significant degree on the temperature and the nature of the cation in the catalyst carrier. Under optimal conditions, each of the enumerated esters can be obtained with the yield of 50–80%.
Similar content being viewed by others
Literature cited
R. Brettle and J. G. Parkin, J. Chem. Soc. C., 1352 (1967).
R. Brettle and D. Seddon, J. Chem. Soc. C., 1153 (1970).
T. Okubo and S. Tsutsumi, Bull. Chem. Soc. Jpn.,37, 1794 (1964).
D. A. White, J. Electrochem. Soc.,124, 1177 (1977).
T. Franklin and T. Honda, Electrochim. Acta,23, 439 (1978).
G. I. Nikishin, M. N. Élinson, and S. K. Fedukovich, Izv. Akad. Nauk SSSR, Ser. Khim., 2154 (1984).
M. N. Élinson, S. K. Fedukovich, and G. I. Nikishin, Izv. Akad. Nauk SSSR, Ser. Khim., 96 (1988).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 11, pp. 2534–2538, November, 1988.
Rights and permissions
About this article
Cite this article
Élinson, M.N., Fedukovich, S.K. & Nikishin, G.I. Electrochemical oxidation of malonic esters in the presence of catalyst carriers — salts of hydriodic acid. Russ Chem Bull 37, 2285–2289 (1988). https://doi.org/10.1007/BF00959879
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00959879