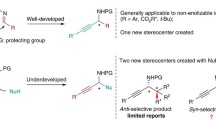
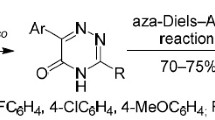
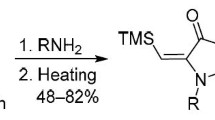
Conclusions
-
1.
Reductive aminolysis of 2-trifluoromethyl-4-benzylidene-Δ2-oxazolin-5-one in the presence of S-amino acid esters, PdCl2 and H2 proceeds via the intermediate formation of RS-peptides.
-
2.
Aminolysis of 2-trifluoromethyl-Δ3-oxazolin-5-ones with S-AlaOMe, S-ValOMe, S-PheOMe proceeds with a high stereoselectivity, with the formation of SS-diastereomer, so that optically pure SS-PheAla and SS-PhePhe dipeptides could be obtained.
-
3.
Aminolysis of 2-trifluoromethyl-Δ3-oxazolin-5-ones with S-ProOMe preferentially gives RS-peptides.
Similar content being viewed by others
Literature cited
Sh. G. Bitiev, G. V. Chel'tsova, E. I. Karpeiskaya, and E. I. Klabunovskii, Izv. Akad. Nauk, Ser. Khim., 2479 (1986).
E. G. Breitholle and G. H. Stammer, J. Org. Chem.,41, 1844 (1976).
M. R. Lyubeznova, K. V. Belyaeva, V. A. Ferapontov, E. I. Karpeiskaya, and E. I. Klavunovskii, Izv Akad. Nauk, Ser. Khim., 2482 (1984).
E. I. Karpeiskaya, L. F. Godunova, E. S. Levitina, M. R. Lyubeznova, E. I. Klabunovskii, E. D. Lubuzh, and A. I. Lutsenko, Izv. Akad. Nauk SSSR, Ser. Khim.,85 (1984).
W. Jenks, Catalysts in Chemistry and Enzymology [Russian translation], Mir, Moscow (1982), p. 350.
E. F. Schallenberg and M. Calvin, J. Am. Chem. Soc.,77, 2779 (1955).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 2, pp. 380–386, February, 1987.
Rights and permissions
About this article
Cite this article
Bitiev, S.G., Chel'tsova, G.V., Lutsenko, M.R. et al. Catalytic asymmetric synthesis of dipeptides from 2-trifluoromethyl-Δ2- and 2-trifluoromethyl-Δ3-oxazolin-5-ones. Russ Chem Bull 36, 339–345 (1987). https://doi.org/10.1007/BF00959378
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00959378